Automatic Fetal Measurements for Low-Cost
Transcription
Automatic Fetal Measurements for Low-Cost
Automatic fetal measurements for low-cost settings by using
Local Phase Bone detection
Benjamin Amoah
1,2 ,
Evelyn Arthur Anto
Abstract— The estimation of gestational age is
done mostly by measurements of fetal anatomical
structures such as the head and femur. These measurement are also used in diagnosis and growth
assessment. Manual measurements is operator dependent and hence subject to variability.
By the proposed method, fetal femurs are automatically measured and gestational ages, hence
delivery dates, are predicted. The initial step is the
detection of biometric elements in fetal ultrasound
scans through local image phase features obtained
by using Gabor filter banks which detect structures
based on the frequency nature of the soft tissue/bone
interfaces. The second step is a regression method to
relate the obtained features to the gestational age.
The lengths of 20 femurs are analyzed in our
experiments. The resulting measurements are consistent with manual measurements obtained by two
expert technicians. The method is fully automatic
and can replace the manual approach to measuring
femur of fetuses in US images.
I. INTRODUCTION
Ultrasound (US) imaging is preferred in obstetrics because it is non-invasive, more economical, safe (since it does not involve any ionizing
radiation) and the images can be produced at
video rate, enabling the observation of the dynamic
behavior of structures over time. The portability
of ultrasound scan machines gives US imaging
an extra advantage. Obstetricians are concerned
about qualitative measurements such as the location, orientation and movement of structures of
interest and quantitative measurements of lengths,
areas and volumes of these structures. Keen among
such measurements are the head circumference,
femur length, bi-parietal diameter and the occipital
frontal diameter. The goals of such measurements
are effective diagnosis and assessment of growth
of foetus. To take such measurements, the contours
of anatomical structures are extracted (segmented).
Manual extraction (which is the current approach
to measuring contours of foetal anatomies) is tedious, time consuming and subject to variability
This work was supported by ETH-Zurich, Switzerland and
African Institute for Mathematical Sciences, Ghana
1 Swiss Federal of Technology (ETH-Zurich)
2 African Institute for Mathematical Sciences, Ghana
1,2 ,
Alessandro Crimi
1,2
in human operators. Variability in measurement
obtained by different operators and even the same
operator at different attempts make the results not
reproducible and hence unreliable.
Little attempts have been made to automate
the extraction of contours in ultrasound images
although a lot have been done with other imaging
modalities. This is, arguable, due to the fact that
ultrasound images are of very poor quality as a result of speckle noise inherent in ultrasound images
[1], [5]. General purpose segmentation approaches
have not been able to deal with the problem.
Previous works involved the use discriminative
constrained probabilistic boosting tree classifiers to
segment structures [3] and iterative approaches to
segment fetal structures by Maximum-Likelihood
estimation [2]. These techniques aim at detecting
in US images, features such as the parietal diameter (BPD), occipital-frontal diameter (OFD),
head circumference (HC) and femur length (FL)
[4]. Recently neuro-developmental maturation of a
fetus based on 3D ultrasound has been introduced
because neurological age-discriminating anatomies
such as the Sylvian fissure, cingulate and callosal
sulci are more indicative than traditional features
[6].
However, all these methods have been tested
mainly in high-end devices which even if considered low-cost in high income countries, they
might not be accessible in low-income countries.
With the goal of improving prenatal care management in rural areas in Ghana, a pilot project has
been carried out in some communities between
the cities of Accra and Cape-Coast [11]. This
involved community health workers together with
US technicians using low-cost portable ultrasound
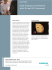
machine as depicted in Fig. 1.
In the following section we report our method,
which is based on a combination of Gabor filtering,
thresholding and structural analysis to automatically segment the femur in US images and to predict gestational age or delivery date. The remainder
of the paper reports the experiments, discussion
and conclusion.
PS(x) =
Fig. 1: Example of ultrasound acquisition in lowcost settings carried out within the project, in a
home of a rural community and not in hospitals or
clinics.
II. METHODS
The method is based on image phase information used for processing US images of hard tissues
such as bones. The phase information is derived by
a bank of Gabor filters (i.e. kernels of Gaussian
functions modulated by sinusoidal plane waves)
applied to a given ultrasound scan. A complex
Gabor filter in 2D is given by (1).
∑r ∑m ||erm (x, y) − |orm (x, y)|| − T
p
, (4)
∑r ∑m erm (x, y)2 + orm (x, y)2 + ε
where ε is a small number to avoid division by zero
and T is a noise threshold. These are dependent on
the specific US machine and found empirically.
The PS image has a maximum at the bone
boundary and hence can be used to identify
the edge of the bone. In our experiments,
we used the parameter values {λ , φ , σ , γ} =
{4.0, 11.0, 1.0, 0.0} which remained constant for
all the testing images. Gabor filters with r = 10
orientations given by θ = nπ
9 , n ∈ {0, 1, 2, . . . , 9}
were used to filter each image. The results of
the PS image were then binarized using an automatic threshold method [10], and then, a dilation followed by an erosion with the structuring
element for the 4-neighborhood are applied. The
used size of the morphological operators was 5.
Although this size was not optimized through the
used dataset, it was noticed that an operator larger
than 10 was compromising the segmentation. The
parameters were obtained base on experimental
results.
A. Femur length
02
2 y02
0
g(x, y, λ , φ , σ , γ) = exp − x +γ
exp 2i π xλ + φ ,
2σ 2
(1)
where {λ , θ , φ , σ , γ} = {wavelength, angle between normal and parallel stripes of the Gabor
function, phase offset, standard deviation, spatial
aspect ratio} and
(x0 , y0 ) = (x cos θ + y sin θ , −x sin θ + y cos θ ).
For simplicity Equation (1) will be written as
g(.). Let Mem (x, y) = real(F −1 (g(.)) and Mom (x) =
imag(F 1 (g(.)) denote the even and odd Log-Gabor
filters at a scale m, with F 1 being the inverse
Fourier transform operation. If the original image
is I(x, y), then the response will be
em (x, y) = I(x, y) ? Mem (x, y),
(2)
Before applying the regression model to predict
the gestational age, small structures are removed.
This was done by using prior knowledge that the
femur is the longest of all structures in a resulting
image. The length of each femur was measured
by the length of the rectangle with minimum area
that encloses all pixels which belong to the femur
contour. The rectangle co-ordinates were computed
automatically [16].
B. Prediction of gestational age
Once the FL is computed, the gestational age
is computed using the Hadlock regression formula used for singleton pregnancies obtained from
mixed population [12], [13]:
GA = 1.863 + 6.280FL − 0.211FL2
(5)
C. Data
and
om (x, y) = I(x, y) ? Mom (x, y).
(3)
Hacihaliloglu et al. [7], [8] argued that a ridge
detector of bone surface is given by a detector
of major axis of symmetry of these transforms.
Therefore a ridge bone detector is given by taking
the difference of these responses over a number of
scales as the measure of phase symmetry (PS) [9],
using different orientation r:
The US scans used were obtained from 20 pregnant women at different stages of pregnancy by using a curvilinear transducer array with 4.5MHz and
a B-Mode US machine (Mindray, Shenzen, China).
All women gave written consent and the study
has been carried out according to the Helsinki
declaration. These images were gray-scale images
with resolution 800 × 600 pixels which were
acquired using different image gain for a better
view, removing labels and menu from the resulting
images. Image acquisition was not carried out in
hospitals or clinics, but in private homes of pregnant women within rural communities in Ghana.
III. E XPERIMENTS AND E VALUATION
The proposed approach was tested on n = 20 different ultrasound images of femurs obtained from
different pregnant women. An example of results
is depicted in Fig. 2, where the original image,
the resulting image and the predicted gestational
age as the final output. A comparison between an
US scan processed using the Canny filter edge
detector and the PS image, before the selection
of the longest segment, is depicted respectively in
Fig.3 and Fig.4.
(±0.09), which signifies substantial agreement between the manual annotations and our automatic
segmentations. A Python script was written to
enable two experts manually measure the length
of the femurs in order to evaluate our automatic
measurements. In Table I, E1 , E2 , and A are
respectively the measurements obtained by the first
expert technician, second expert technician and
our automatic measurements. Ē, which represents
the actual manual measurement of the femur, is
the average of E1 and E2 ; d1 = (E1 − E2 )2 and
d2 = (Ē − A)2 . The mean of d2 is 0.03. A paired
t-test (two-tailed p-value equals 0.1165) shows a
difference is not statistically significant, thus, the
measurements by the our approach are at least
as good as the manual measurements. Moreover,
the estimation error (0.03) using the automatic
approach is significantly (p < 0.01) lesser than the
inter-expert variability (0.11). These show that our
simple approach gives accurate measurements and
can replace the manual system.
TABLE I: Results from Experiments
Fig. 3: Example of ultrasound scan processed using
Canny filter edge detector.
Fig. 4: Example result of the PS image before the
selection of the longest edge as a femur.
To assess the accuracy of our approach, a kappastatistic (κ) [17] is calculated for each of the 20
segmented images. A careful manual segmentation of each image is used as the ground truth
segmentation. The κ-values obtained (as shown
in Table I) ranged has mean (±SD) being 0.74
No.
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
Avg
SD
κ
0.66
0.62
0.73
0.80
0.82
0.76
0.61
0.82
0.83
0.76
0.63
0.80
0.84
0.73
0.73
0.49
0.77
0.72
0.85
0.81
0.74
0.09
E1
5.10
4.90
4.57
3.41
3.16
5.18
4.98
4.07
3.86
3.99
4.63
4.14
2.65
3.92
4.11
4.44
4.07
2.63
2.65
2.50
4.02
0.84
E2
4.84
4.69
4.13
3.23
3.08
5.05
4.61
3.90
3.72
3.54
4.04
4.31
2.56
3.57
3.50
4.25
3.43
2.38
2.51
2.46
3.69
0.80
Ē
4.97
4.80
4.35
3.32
3.12
5.12
4.80
3.99
3.79
3.77
4.34
4.23
2.61
3.75
3.81
4.35
3.75
2.51
2.58
2.48
3.82
0.81
A
4.97
4.96
4.29
3.53
3.23
5.00
4.67
4.11
3.77
3.97
4.23
4.23
2.30
3.48
3.55
4.16
3.55
2.39
2.41
2.43
3.76
0.85
d1
0.07
0.04
0.19
0.03
0.01
0.02
0.14
0.03
0.02
0.20
0.35
0.03
0.01
0.12
0.37
0.04
0.41
0.06
0.02
0.00
0.11
0.13
d2
0.00
0.03
0.00
0.04
0.01
0.01
0.02
0.02
0.00
0.04
0.01
0.00
0.09
0.07
0.07
0.03
0.04
0.01
0.03
0.00
0.03
0.03
The Gestational age predicted with our method
allowed the women to be better prepared for their
delivery days, hence, reducing the number of home
deliveries [11].
IV. DISCUSSIONS
Rather than obtaining a perfect segmentation of
the femur, the goal is to obtain a bounding box
with length as close as possible to femur lengths
obtained by sonographers. This approach has been
tested using low-cost settings such as low-cost
Fig. 2: From left to right: original image, segmented femur, and predicted gestational age.
portable ultrasound and image acquisition under
sub-optimal conditions.With our fully automatic
approach which gives consistent measurements,
the issue of operator dependent femur length acquisition is addressed.
Future works include the analysis of other biometric features linked to the gestational age and
the correlation to the weight to assess the wellbeing of the fetus [15]. Regarding the gestational
age prediction, some authors argue that there is
no evidence that ethnicity significantly influence
the measurements, though no agreement has been
reached [14]. A larger study with bigger population
can prove the validity of the proposed approach for
low-income countries in West-Africa.
V. CONCLUSIONS
The proposed approach integrating several image processing steps obtain the femur length and
subsequently estimate the gestational age, to allow user-independent fetal feature assessment. In
particular, we investigated the feasibility of the
method in low-cost settings and on a small WestAfrican population. The method is fully automatic
and requires little training to be used by physicians
and/or sonographers.
ACKNOWLEDGMENT
The authors are thankful to Dr. Kojo Pieterson for the clinical support. This research is part
of the cooperation project carried out in rural
communities in Ghana called Docmeup by ETHGlobal and the African Institute for Mathematical
Sciences in Ghana [11]. More resources related to
the cooperation project can be found on the website
www.docmeup.org.
R EFERENCES
[1] S. Kalpana, M. L. Dewal, and M. Rohit, ”Ultrasound
imaging and image segmentation in the area of ultrasound:
a review,” International Journal of Advanced Science and
Technology, vol. 24, Nov. 2010.
[2] S.M. Jardim, and M.A. Figueiredo, ”Segmentation of fetal
ultrasound images,” Ultrasound in medicine & biology,
vol. 31, no. 2, 2005, 243-250.
[3] G. Carneiro and B. Georgescu and S. Good and D. Comaniciu, ”Detection and measurement of fetal anatomies
from ultrasound images using a constrained probabilistic
boosting tree,” IEEE Trans Med. Imaging, vol. 27, no. 9,
1342-1355, Sept. 2008.
[4] S. Rueda and et al.”Evaluation and comparison of current
fetal ultrasound image segmentation methods for biometric measurements: a grand challenge,” Medical Imaging,
IEEE Transactions on, vol. 33, 797-813.
[5] J.A. Noble and D. Boukerroui. ”Ultrasound image segmentation: a survey.” Medical Imaging, IEEE Transactions on 25.8 (2006): 987-1010.
[6] A. Namburete and et al. ”Learning-based prediction of
gestational age from ultrasound images of the fetal brain.”
Medical image analysis 21.1 (2015): 72-86.
[7] I. Hacihaliloglu and et al. ”Bone surface localization in
ultrasound using image phase-based features.” Ultrasound
in medicine and biology 35.9 (2009): 1475-1487.
[8] I. Hacihaliloglu and et al. ”Bone segmentation and fracture detection in ultrasound using 3D local phase features.” Medical Image Computing and Computer-Assisted
InterventionMICCAI 2008. Springer Berlin Heidelberg,
2008. 287-295.
[9] P. Kovesi ”Symmetry and Asymmetry from Local Phase”
Proc Tenth Australian Joint Conference on Artificial Intelligence (1997), 185-190
[10] N. Otsu, ”A threshold selection method from gray-level
histograms.” Automatica 11.285-296 (1975): 23-27.
[11] B. Amoah and E.A. Anto and A. Crimi, Phone-based
prenatal care for communities and remote ultrasound
imaging, in MobMed Prague, 2014.
[12] F. Varol, et al. ”Evaluation of gestational age based on
ultrasound fetal growth measurements.” Yonsei medical
journal 42.3 (2001): 299-303.
[13] F. Hadlock, et al. ”Estimating fetal age: computer-assisted
analysis of multiple fetal growth parameters.” Radiology
152.2 (1984): 497-501.
[14] L.K. Pemberton and I. Burd and E. Wang. ”An appraisal
of ultrasound fetal biometry in the first trimester.” Reports
in Medical Imaging 3 (2010): 11-15.
[15] N.J. Secher and et al. ”Estimation of fetal weight in
the third trimester by ultrasound.” European Journal of
Obstetrics and Gynecology and Reproductive Biology
24.1 (1987): 1-11.
[16] D. Chaudhuri and A. Samal. ”A simple method for
fitting of bounding rectangle to closed regions.” Journal
of Pattern Recognition 40 (2007): 1981-1989.
[17] A.J. Viera and J.M. Garrett. ”Understanding interobserver agreement: the kappa statistic.” Family Medicine
2005;37(5):360-3