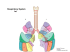Approach to HRCT Diagnosis and Findings of Lung Disease
Transcription
Approach to HRCT Diagnosis and Findings of Lung Disease
S E C T I O N II Approach to HRCT Diagnosis and Findings of Lung Disease The high-resolution computed tomography (HRCT) detection and diagnosis of diffuse lung disease is primarily based on the recognition of (a) a limited number of specific abnormal findings, (b) specific combinations or patterns of these abnormalities, (c) one or more specific distributions of abnormal findings, and (d) the use of basic history and clinical information. HRCT FINDINGS OF LUNG DISEASE Abnormal HRCT findings, which have been enumerated over the last 25 years, and their differential diagnosis are reviewed in the subsequent five chapters. These findings can be classified in general terms as: 1. linear and reticular opacities; 2. multiple nodules and nodular opacities; 3. parenchymal opacification, including consolidation and ground-glass opacity; 4. air-filled cystic lesions, including lung cysts, cystic lung disease, emphysema, and dilated bronchi (bronchiectasis); and 5. decreased lung attenuation, including mosaic perfusion, mosaic attenuation, and air trapping on expiratory scans. (Continued) #152201 Cust: LWW Au: Webb Pg. No. 71 DESIGN SERVICES OF COMBINATIONS AND PATTERNS OF HRCT ABNORMALITIES The diagnosis or differential diagnosis of diffuse lung disease is often based on the recognition of specific combinations of HRCT abnormalities, representing specific patterns of disease. For example, in a patient with honeycombing visible on HRCT, idiopathic pulmonary fibrosis may be a likely diagnosis, but if honeycombing is associated with mosaic perfusion or air trapping, hypersensitivity pneumonitis should be suggested instead. On the other hand, mosaic perfusion or air trapping associated with bronchiectasis suggests airways disease. Furthermore, in some patients, ancillary abnormalities such as lymph node enlargement, mediastinal and cardiac abnormalities, or abnormalities in the upper abdomen may be helpful. DISTRIBUTION OF HRCT ABNORMALITIES When attempting to reach a diagnosis or differential diagnosis of lung disease using HRCT, the predominant distribution of abnormalities must be considered along with their appearance and morphology, and the combination or pattern or abnormal findings present. Although abnormalities in patients with a diffuse lung disease may involve the entire lung to an equal degree, a specific predominance in one or more regions is often discernable. Many lung diseases show specific regional distributions, or a predominance in relation to specific lung structures, a fact that is related to their underlying histology, pathogenesis, and pathophysiology. In different diseases, abnormalities may predominate in relation to: 1.one lung. Many lung diseases are diffuse, and involve both lungs to an equal or nearly equal degree. On the other hand, some diseases may be asymmetrical, predominating in one lung, or may show this finding in some cases. A few lung diseases can be unilateral. 2.the lung in cross section, as displayed on transaxial HRCT images. In different diseases, abnormalities may predominate in (a) the peripheral or subpleural lung, (b) the lung periphery, but with relative sparing of the immediate subpleural regions, or (c) central or peribronchovascular regions, sparing the supleural lung, or maybe (d) diffuse, equally involving the entire cross section of lung. 3.the upper-, mid-, or lower lungs. This predominance may be ascertained by comparing the severity of abnormal findings on transaxial scans through the upper-, mid-, and lower lung regions, or by using two-dimensional reconstructions from volumetric imaging. 4.the anterior or posterior lung as seen on transaxial images or sagittal reconstructions. 5.the secondary pulmonary lobule or lobular structures, being centrilobular, bronchiolar, perilobular, involving the interlobular septa, or lobular. 6.specific lung structures, such as the pleura (visceral or parietal), bronchi, or vascular structures, or a combination of specific lung structures. For example, some patients with nodular lung disease may show a preponderance of nodules in relation to bronchi and peribronchovascular regions, the subpleural lung, and interlobular septa; this combination is termed a lymphatic or perilymphatic distribution. It is typical of sarcoidosis and a few other diseases. It is important to keep in mind that predominance in more than one of the regions described above may be identified in any given case; as when identifying specific HRCT abnormalities, a specific combination of these may be suggestive of a particular diagnosis or differential diagnosis. For example, in a patient with sarcoidosis, a perilymphatic distribution of nodules on HRCT is usually associated with upper-lobe predominance, and the abnormalities may be symmetrical or asymmetrical. Also, significant variations in classical patterns of lung involvement can be seen in individual patients. A specific diagnosis that otherwise seems likely should not be excluded because of an atypical distribution of abnormalities. 72 CLINICAL FINDINGS Although history and other clinical findings can be of great value in suspecting or diagnosing a specific disease, in clinical practice, many HRCT studies are performed and interpreted with little or no clinical information available. Patients may be referred for HRCT without having seen a local physician or pulmonologist, or prior to their first appointment. However, even in such cases, some basic clinical information useful in diagnosis is often available. Such basic history as whether the patient’s symptoms are acute or chronic, or whether a fever is present, can be helpful. These will be stressed in the subsequent chapters, as specific findings and patterns are reviewed. For example, in a patient with HRCT showing ground-glass opacity as the predominant HRCT abnormality, knowing whether symptoms are acute or chronic can limit an otherwise lengthy and nonspecific differential diagnosis. In a patient with ground-glass opacity and acute symptoms, the most likely diagnoses include pulmonary edema or hemorrhage, atypical pneumonia, aspiration, or diffuse alveolar damage; in a similar patient with progressive or chronic symptoms, the differential diagnosis is long, and includes such diseases as hypersensitivity pneumonitis, nonspecific interstitial pneumonia (NSIP), desquamative interstitial pneumonia (DIP), respiratory bronchiolitis-interstitial lung disease (RB-ILD), lymphoid interstitial pneumonia (LIP), organizing pneumonia (OP), eosinophilic lung disease, alveolar proteinosis, lipoid pneumonia, and invasive pulmonary mucinous adenocarcinoma. 73 3 HRCT Findings: Linear and Reticular Opacities I M P O R T A N T T O P I C S INTERLOBULAR SEPTAL THICKENING 74 HONEYCOMBING 82 INTRALOBULAR INTERSTITIAL THICKENING (INTRALOBULAR LINES) 89 PARENCHYMAL BANDS 97 NONSPECIFIC RETICULATION 92 SUBPLEURAL CURVILINEAR LINE 98 THE INTERFACE SIGN 92 NORMAL RETICULAR OPACITIES IN THE ELDERLY 100 TRACTION BRONCHIECTASIS AND TRACTION BRONCHIOLECTASIS 93 Abbreviations Used in This Chapter AIP ARDS COPD CWP DAD DIP HP IP IPF LIP NSIP OP RB-ILD UIP PERIBRONCHOVASCULAR INTERSTITIAL THICKENING 94 acute interstitial pneumonia acute respiratory distress syndrome chronic obstructive pulmonary disease coal worker’s pneumoconiosis diffuse alveolar damage desquamative interstitial pneumonia hypersensitivity pneumonitis interstitial pneumonia idiopathic pulmonary fibrosis lymphoid interstitial pneumonia nonspecific interstitial pneumonia organizing pneumonia respiratory bronchiolitis-interstitial lung disease usual interstitial pneumonia Thickening of the lung interstitium by fluid, fibrous tissue, or because of cellular infiltration usually results in an increase in reticular or linear opacities on high-resolution computed tomography (HRCT). Reticular abnormalities identifiable on HRCT are generally characterized as belonging to one of three recognizable patterns, which can sometimes be seen together. These are (a) interlobular septal thickening, (b) honeycombing, and (c) intralobular interstitial thickening, also described as intralobular lines by its HRCT appearance (Fig. 3-1). The first two of these are most easily recognized and have a limited differential diagnosis. The last is less specific. SUBPLEURAL INTERSTITIAL THICKENING 98 DISTRIBUTION OF RETICULAR OPACITIES IN THE DIAGNOSIS OF LUNG DISEASE 100 Additional findings that may be seen in isolation or associated with one or more of these reticular patterns include traction bronchiectasis, the interface sign, peribronchovascular interstitial thickening, parenchymal bands, subpleural interstitial thickening, and subpleural lines (Fig. 3-1). Interlobular Septal Thickening On HRCT, numerous clearly visible interlobular septa almost always indicate the presence of an interstitial abnormality; only a few septa should be visible in normal patients (see Chapter 2). Septal thickening can be seen in the presence of interstitial fluid, cellular infiltration, infiltration by other materials such as amyloid, lymphatic dilatation or proliferation, or fibrosis. Interlobular septal thickening can be diagnosed if visible linear opacities can be seen to outline what can be recognized as pulmonary lobules because of their characteristic size and shape. Within the peripheral lung, thickened septa 1 to 2 cm in length may outline part of or an entire lobule and are usually seen extending to the pleural surface, being roughly perpendicular to the pleura (Figs. 3-1 to 3-14) (1–8). Lobules at the pleural surface may have a variety of appearances, but they are often longer than they are wide, resembling a cone or truncated cone. Within the central lung, thickened septa usually outline lobules that are 1 to 2.5 cm in diameter and appear polygonal, or sometimes hexagonal, in shape (Fig. 3-2). Lobules delineated by thickened septa commonly contain a visible dotlike or branching centrilobular pulmonary 74 #152201 Cust: LWW Au: Webb Pg. No. 74 DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 75 Interlobular septal thickening Parenchymal bands Centrilobular peribronchovascular interstitial thickening Peribronchovascular interstitial thickening Intralobular interstital thickening Fissure thickening due to subpleural interstitial thickening Traction bronchiectasis Peripheral honeycombing Honeycombing producing a subpleural line Bronchiectasis “signet ring sign” Subpleural line without honeycombing FIGU RE 3-1 Linear and reticular opacities visible on HRCT. A B FIGU RE 3-2 Smooth interlobular septal thickening in two patients with pulmonary edema. A: The reticular pattern can be recognized as interlobular septal thickening because the lines outline recognizable pulmonary lobules. A lobule in the anterior lung outlined by interlobular septa (yellow arrows) shows dotlike pulmonary artery branches in its center (red arrows). Small nodular opacities (blue arrows) seen in relation to some septa represent pulmonary vein branches seen in cross section. B: Smooth thickening of numerous interlobular septa is visible in the upper lobe. Smooth peribronchovascular interstitial thickening (appearing as peribronchial cuffing or bronchial wall thickening) is also present. This finding is commonly associated with interlobular septal thickening. #152201 Cust: LWW Au: Webb Pg. No. 75 DESIGN SERVICES OF 76 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease FIGURE 3-3 Smooth interlobular septal thickening in a patient with pulmonary edema. A coronal reconstruction shows smoothly thickened interlobular septa (arrows), which are most evident in the peripheral lung. Thickening of the peribronchovascular interstitium and subpleural interstitial thickening are present. artery. The characteristic relationship of the interlobular septa and centrilobular artery is often of value in identifying each of these structures. The terms septal lines or septal thickening (Figs. 3-1 to 3-14) may also be used to describe interlobular septal A FIGURE 3-4 Smooth interlobular septal thickening in a patient with lymphangitic carcinoma. This appearance is indistinguishable from pulmonary edema. thickening (9,10), and these terms are preferred to earlier descriptions such as peripheral lines, short lines, and interlobular lines (4,8,11). Similarly, although thickened septa outlining one or more pulmonary lobules have been described as producing a “large reticular B FIGU RE 3-5 A–C: Interlobular septal thickening in a patient with lymphangitic spread of breast carcinoma. Diffuse, smooth interlobular septal thickening outlines numerous pulmonary lobules, primarily in the right lung. In addition to septal thickening, there is increased prominence of the peribronchovascular interstitium, mostly easily recognized as bronchial wall thickening (B, arrow). A small pneumothorax is visible on the right because of a recent thoracentisis. #152201 Cust: LWW Au: Webb Pg. No. 76 C DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 77 FIGU RE 3-6 Smooth interlobular septal thickening in a child with lymphangiomatosis. Mediastinal widening is also present. FIGU RE 3-8 Interlobular septal thickening in alveolar proteinosis. Thickened septa are associated with ground-glass opacity. The combination of interlobular septal thickening and ground-glass opacity in the same lung region is typical of alveolar proteinosis and is termed crazy paving. pattern” (1,12) or “polygons” (13), and, if they can be seen contacting the pleural surface, as “peripheral arcades” or “polygonal arcades” (4), the terms interlobular septal thickening, septal thickening, and septal lines are considered more specific in describing these appearances (9,14). Thickening of the interlobular septa is commonly seen in patients with interstitial lung diseases (15,16), but may also be seen in normal elderly patients (17) and otherwise normal smokers. The presence of septal thickening is of little diagnostic value when other HRCT abnormalities are also visible (15). However, when visible as an isolated or predominant abnormality, this finding has a limited differential diagnosis (Table 3-1). Septal thickening can be smooth, nodular, or irregular in contour in different pathologic processes (2,18–21). A simple algorithm (Fig. 3-15) based on the recognition of these findings may be useful for diagnosis. Regardless of the cause or appearance of septal thickening, this finding is often associated with peribronchial interstitial thickening and subpleural interstitial thickening, which are described later. A Smooth Septal Thickening B FIGU RE 3-7 Smooth interlobular septal thickening in Erdheim–Chester disease, a non-Langerhans cell histiocytosis that can result in lung infiltration along lymphatics. A: Thickening of numerous interlobular septa is visible on HRCT. B: Sagittal lung slice after lung removal for transplantation. Thickened interlobular septa are most evident in the upper lobes. Histology showed a combination of fibrosis and histiocytic infiltration. (Courtesy of Kevin O. Leslie MD, Mayo Clinic, Scottsdale.) #152201 Cust: LWW Au: Webb Pg. No. 77 Smooth septal thickening is usually seen in patients with venous, lymphatic, or infiltrative diseases (19). Specifically, it may be seen in the presence of pulmonary edema or hemorrhage (Figs. 3-2 and 3-3) (22–25), pulmonary veno-occlusive disease (22,24,26,27), lymphangitic spread of carcinoma (Figs. 3-4 and 3-5) (4,7,28), lymphoma, DESIGN SERVICES OF A B FIGU RE 3-9 “Beaded” or nodular septal thickening in two patients with sarcoidosis. A: Interlobular septa in the upper lobe are nodular in appearance (arrows); this has been termed the beaded septum sign. B: Numerous nodules are visible within interlobular septa (arrows). The nodules are too numerous to represent normal pulmonary veins. A B FIGURE 3-10 Nodular interlobular septal thickening in C D lymphangitic spread of carcinoma. A–C: Nodular interlobular septal thickening in a patient with metastatic colon carcinoma. Nodules are clearly visible within the septa outlining a lobule in the lung apex (arrows, A). D: Nodular septal thickening (arrows) shown in a lung specimen of a patient with lymphangitic spread of carcinoma. 78 #152201 Cust: LWW Au: Webb Pg. No. 78 DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 79 A FIGU RE 3-12 Irregular interlobular septal thickening in patients with fibrotic sarcoidosis. Numerous irregularly thickened interlobular septa (arrows) are associated with lung fibrosis and distortion of lung architecture. Some nodules remain visible. (34), some pneumonias (35), and in a small percentage of patients with pulmonary fibrosis. Smooth interlobular septal thickening, regardless of its cause, is often associated with smooth peribronchovascular and subpleural interstitial thickening, which is most easily recognized as thickening of fissures. As discussed below, perilobular abnormalities in patients with organizing pneumonia (OP) can mimic smooth interlobular septal thickening (Fig. 3-14) (36). In many diseases associated with smooth septal thickening, the thickening is diffuse. The primary exception is lymphangitic spread of neoplasm, in which the abnormality may be unilateral or bilateral, asymmetrical, patchy, and upper- or lower lobe predominant. Also, in patients with diffuse septal thickening, or conditions such as pulmonary edema, in which a basal predominance may be seen, often thickened septa are best defined in the apices and upper lobes, as interlobular septa are best developed in this region. Smooth septal thickening may also be seen in association with ground-glass opacity, a pattern termed crazy paving (see Chapter 5). This pattern is typical of alveolar proteinosis (Fig. 3-8) but has a long differential diagnosis, which is reviewed in Chapter 5 (37–42). B C FIGU RE 3-11 Irregular interlobular septal thickening in patients with fibrotic HP. A and B: Irregular interlobular septal thickening is visible (arrows) in less-abnormal regions, but in areas with severe fibrosis, septa are more difficult to recognize. C: In a different patient with chronic HP with fibrosis, well-defined septa are visible in less-abnormal regions at the lung bases (red arrows), but are more difficult to recognize and more irregular in contour in more severely involved regions (yellow arrows). leukemia, and lymphoproliferative diseases; lymphangiomatosis (Fig. 3-6) (29,30); congenital pulmonary lymphangiectasia (31,32), interstitial infiltration associated with amyloid (33), Erdheim-Chester disease (Fig. 3-7) #152201 Cust: LWW Au: Webb Pg. No. 79 Nodular Septal Thickening Nodular or “beaded” septal thickening occurs in l ymphatic or infiltrative diseases, including lymphangitic spread of carcinoma and lymphoma (4,7,28), lymphoproliferative disease such as lymphoid interstitial pneumonia (LIP) (43–45), sarcoidosis (46–49), silicosis or coal worker’s pneumoconiosis (CWP) (50), and amyloidosis or lightchain deposition disease (33,51) (Figs. 3-9 and 3-10). Nodular septal thickening is most appropriately considered along with other nodular patterns of diffuse lung disease. Septal nodules are often associated with a so-called “perilymphatic” or “lymphatic” distribution of nodules, in DESIGN SERVICES OF 80 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease A B FIGU RE 3-13 Irregular septal thickening in UIP. A: Irregular reticular opacities (arrows) are visible in the peripheral lung in a patient with pulmonary fibrosis related to treatment with methotrexate. These may represent irregularly thickened septa or perilobular fibrosis. B: Irregular interlobular septal thickening or perilobular fibrosis (arrows) in a patient with IPF. C: Histologic section in a patient with IPF. Irregular bands of fibrosis (arrows) are visible within the periphery of lobules, involving the interlobular septa. C A B FIGU RE 3-14 A perilobular pattern in OP. A and B: Thickened interlobular septa and thicker arcades are visible (arrows) in a patient with OP related to dematomyositis. Areas of consolidation are also present. #152201 Cust: LWW Au: Webb Pg. No. 80 DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 81 TABLE 3-1 Differential Diagnosis of Interlobular Septal Thickening Diagnosis Comments Lymphangitic carcinomatosis, lymphoma, leukemia Common; predominant finding in most; usually smooth; sometimes nodular Lymphoproliferative disease (e.g., LIP) Smooth or nodular; other abnormalities (i.e., nodules) typically present Lymphangiomatosis Rare, smooth Congenital pulmonary lymphangiectasia Rare, smooth Pulmonary edema Common; predominant finding in most; smooth; ground-glass opacity can be present Pulmonary hemorrhage Smooth; associated with ground-glass opacity Erdheim-Chester disease Rare, smooth Pneumonia (e.g., viral, Pneumocystis carinii) Smooth; associated with ground-glass opacity Sarcoidosis Common; usually nodular or irregular; conglomerate masses of fibrous tissue with traction bronchiectasis typical in end stage IPF or other cause of UIP Sometimes visible but not common; appears irregular; intralobular thickening and honeycombing usually predominates NSIP With findings of ground-glass opacity and reticulation Silicosis/CWP; talcosis Occasionally visible; usually nodular; irregular in end-stage disease Asbestosis Sometimes visible; irregular HP (chronic) Uncommon; irregular reticular opacities and honeycombing usually predominate Amyloidosis Smooth or nodular OP Perilobular pattern; thick, ill-defined “septal thickening” Elderly patients Some septal thickening normal which abnormalities occur primarily in relation to pulmonary lymphatics (9,14,30,47). In addition to septal nodules, a perilymphatic pattern is associated with interstitial thickening or nodules involving (a) the subpleural regions, (b) the peribronchovascular interstitium in a perihilar location, and (c) the centrilobular peribronchovascular interstitium. This pattern is most typical of patients with sarcoidosis, silicosis, lymphangitic spread of carcinoma or other neoplasms, and lymphoproliferative disease. Nodular patterns of diffuse lung disease are discussed in detail in the next chapter. Nodular septal thickening in sarcoidosis, silicosis, and CWP is usually best seen in the upper lobes and parahilar regions, because of the tendency of these diseases to predominate in the upper lobes, but this is not always the case. Lymphoproliferative disease is often diffuse in distribution or basal predominant. Lymphangitic spread Interlobular septal thickening Irregular (lung distortion) Smooth Thick septa predominant Sarcoidosis Asbestosis UIP Fibrotic HP Edema Lymphangitic tumor Lymphoproliferative disease Lymphangiectasia Amyloidosis Pulmonary veno-occlusive disease Erdheim-Chester disease OP Nodular Ground-glass opacity predominant “Crazy-paving” differential diagnosis Perilymphatic diseases; Sarcoidosis Lymphangitic tumor Lymphoproliferative disease Silicosis and CWP Amyloidosis FIGU RE 3-15 Algorithmic approach to the diagnosis of interlobular septal thickening. #152201 Cust: LWW Au: Webb Pg. No. 81 DESIGN SERVICES OF 82 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease of neoplasm has a variable distribution, and may be diffuse or localized. Irregular Septal Thickening In patients who have interstitial fibrosis, septal thickening visible on HRCT is often irregular in appearance and associated with distortion of lung architecture (Figs. 3-11 to 3-13) (52–56). Although interlobular septal thickening can be seen on HRCT in association with fibrosis and honeycombing (11), it is not usually a predominant feature (5,57,58). Generally speaking, in the presence of significant fibrosis and honeycombing, distortion of lung architecture makes the recognition of thickened septa difficult, except in less-involved lung regions (Fig. 3-11). Among patients with pulmonary fibrosis and “end-stage” lung disease, the presence of interlobular septal thickening on HRCT is most frequent in patients with sarcoidosis (Fig. 3-12) (56% of patients) and is less common in those with usual interstitial pneumonia (UIP) of various causes (Fig. 3-13), asbestosis, and hypersensitivity pneumonitis (HP) (Fig. 3-11) (58). The frequency of septal thickening and fibrosis in patients with sarcoidosis reflects the tendency of active sarcoid granulomas to involve the interlobular septa. In patients with idiopathic pulmonary fibrosis (IPF) or UIP of other cause, irregular reticular opacities are often visible on HRCT, which appear to represent thickened interlobular septa. However, this finding usually correlates with the presence of fibrosis predominantly affecting the periphery of acini and the secondary lobule rather than the septa themselves (30,57). Nonetheless, the HRCT appearance is similar to that of irregular septal thickening (Fig. 3-13). In patients with irregular interlobular septal thickening resulting from fibrosis, other findings such as honeycombing, traction bronchiectasis, and the distribution of abnormalities are usually most valuable in differential diagnosis. The Perilobular Pattern Pulmonary disease occurring predominantly in relation to interlobular septa and the periphery of lobules has been termed perilobular (10,19,36,59,60). Johkoh et al. (41,60) emphasized that a perilobular distribution of disease may reflect abnormalities of the peripheral alveoli and subpleural interstitium in addition to thickening of interlobular septa (Fig. 3-13). Peripheral lobular fibrosis may result in irregular reticular opacities, which mimic the appearance of interlobular septal thickening. A peripheral lobular or “perilobular” distribution of abnormalities has been reported in as many as half of patients with OP (36). These abnormalities result in an appearance (on HRCT) of arcuate or polygonal opacities, which are less well defined, and may be thicker, than thickened interlobular septa, and may be associated with areas of ground-glass opacity or consolidation #152201 Cust: LWW Au: Webb Pg. No. 82 (Fig. 3-14). Although the histologic correlates of this pattern are unclear, it is likely related to OP involving distal airspaces. HONEYCOMBING Extensive interstitial fibrosis that results in alveolar disruption and bronchiolectasis produces the classic and characteristic appearance of honeycombing or honeycomb lung (61). Pathologically, honeycombing is defined by the presence of small air-containing cystic spaces, generally lined by bronchiolar epithelium and having thickened walls composed of dense fibrous tissue. Honeycombing indicates the presence of end-stage lung and can be seen in a number of diseases leading to end-stage pulmonary fibrosis (58,62). Honeycombing produces a characteristic cystic appearance on HRCT, and when present, allows a confident diagnosis of lung fibrosis (Table 3-2) (5,52,61). On HRCT, the cystic spaces of honeycombing usually range from 3 mm to 1 cm in diameter, although they can be as large as several centimeters in diameter; they are characterized by clearly definable walls 1 to 3 mm in thickness (5,52) (Figs. 3-1 and 3-16 to 3-19). The cysts are air-filled and appear lucent in comparison to normal lung parenchyma. Although there is some overlap between the appearances of fine honeycombing and intralobular interstitial thickening, if the spaces between the lines (i.e., the holes) appear to be air-filled (i.e., black), rather than having the density of lung parenchyma, honeycombing is likely present. Honeycombing has been described by Zerhouni et al. as producing an “intermediate reticular pattern” to distinguish it from the larger pattern seen with interlobular septal thickening and the smaller pattern visible with intralobular interstitial thickening (12). Honeycomb cysts often predominate in the peripheral and subpleural lung regions regardless of their cause, and perihilar lung can appear normal despite the presence of extensive peripheral abnormalities (Fig. 3-16). It must be emphasized that unless cysts are visible in the immediate subpleural lung, honeycombing cannot be diagnosed with certainty. Airfilled cysts that are not subpleural may represent traction bronchiectasis, emphysema, pneumatoceles, or a cystic lung disease such as lymphangiomyomatosis or Langerhans cell TABLE 3-2 HRCT Characteristics of Honeycomb Cysts Thick, easily seen walls Air-filled (i.e., black) Usually 3–10 mm in diameter Immediately subpleural in location They occur in clusters or layers and share walls (multiple layers are seen in late disease) Nonbranching Associated with other findings of fibrosis (traction bronchiectasis, irregular reticulation, volume loss, lung distortion) DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities A B FIGU RE 3-16 A — C: Honeycombing in IPF. A: HRCT C A shows honeycomb cysts in the peripheral and subpleural regions. They are air-filled and have a thick and easily recognizable wall. Note that the cysts occur in several layers and are generally less than 1 cm in diameter. B: Resected left lung at a similar level in a different patient with IPF shows honeycomb cysts, which are most extensive in the posterior and peripheral lung. C: Sagittal lung slice in a patient with IPF shows honeycombing (arrows) in the posterior subpleural lung. (Courtesy of Martha Warnock, MD.) B FIGU RE 3-17 A and B: Honeycombing in a patient with IPF (prone HCRT). Honeycombing results in cysts of various sizes, which have a peripheral predominance. The cysts have thick and clearly defined walls. In areas of honeycombing, lobular anatomy cannot be resolved because of architectural distortion. #152201 Cust: LWW Au: Webb Pg. No. 83 DESIGN SERVICES OF 83 A B FIGU RE 3-18 A–C: Honeycombing in rheumatoid lung disease. HRCT shows honeycomb cysts with a distinct subpleural predominance. The cysts are generally smaller than 1 cm in diameter and share walls. Other findings of fibrosis include irregular thickening of the left major fissure (B, arrow) and traction bronchiectasis (C, arrow). C A B FIGURE 3-19 Honeycombing in association with paraseptal emphysema in a patient with IPF. A: Some cysts in the lung periphery, particularly in the left lung, likely reflect paraseptal emphysema, rather than lung fibrosis. The cysts are larger than 1 cm. B and C: More typical honeycombing is visible in the posterior right lung base. Emphysema and honeycombing may occur in combination in some patients, and their distinction on any one slice may be difficult. Emphysema, however, predominates in the upper lobes and honeycombing predominates in the lower lobes. In the presence of emphysema, honeycomb cysts may be larger than is typical. C 84 #152201 Cust: LWW Au: Webb Pg. No. 84 DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities TABLE 3-3 Comparison of HRCT Features of Paraseptal Emphysema and Honeycombing Paraseptal Emphysema Honeycombing Layers One layer One or more layers Associated findings Centrilobular emphysema, bullae in some Traction bronchiectasis, reticulation Distribution Upper-lobe predominant Lower-lobe predominant in most Size Often >1 cm Usually <1 cm Overall lung volume Increased Decreased histiocytosis. Also, a basal predominance is usually present, a finding that is helpful in distinguishing honeycombing from paraseptal emphysema, in which subpleural cysts are visible. In early honeycombing, only a few isolated subpleural cysts may be seen, but it is best to reserve a diagnosis of honeycombing for scans showing clusters, groups, or rows of clearly defined subpleural cysts with easily recognized walls (Figs. 3-16 to 3-18). It is generally a good idea to be conservative when describing this finding as it means that pulmonary fibrosis is present, and it is an essential criterion in the diagnosis of UIP and IPF (10,63). A reasonable, but admittedly arbitrary, rule of thumb would be that honeycombing can be diagnosed if at least three air-filled (black) cysts, 3 to 10 mm in diameter, with thick, recognizable walls, are seen in a row or cluster in a subpleural location (Fig. 3-17). Extensive subpleural honeycomb cysts share walls and often occur in several contiguous layers (Figs. 3-16 to 3-18). This latter finding can allow honeycombing to be distinguished from subpleural emphysema (paraseptal emphysema); in paraseptal emphysema, subpleural cysts usually occur in a single layer (Table 3-3). But also keep in mind that paraseptal emphysema and honeycombing may coexist; in such patients, the cystic spaces of honeycombing may appear larger than usual, and it may be difficult to determine where emphysema stops and honeycombing begins (Fig. 3-19). The differentiation of honeycombing and paraseptal emphysema is further discussed in Chapter 6. Lung consolidation or infiltration in a patient with emphysema or cystic lung disease can mimic the appearance of honeycombing. Honeycombing is usually associated with other findings of lung fibrosis, such as volume loss, architectural distortion, intralobular lines, traction bronchiectasis, traction bronchiolectasis, and irregular subpleural interstitial thickening. Subpleural cysts seen in the absence of other findings of fibrosis likely represent another abnormality such as emphysema. Significant interlobular septal thickening is not commonly visible in association with honeycombing, except in patients with sarcoidosis (58). In patients with HRCT findings of septal thickening, the presence of honeycombing distinguishes fibrosis from other causes of reticulation, such as pulmonary edema or lymphangitic spread of carcinoma. Significance of Honeycombing The presence of honeycombing on HRCT is indicative of significant lung fibrosis and, in many cases, will lead to a diagnosis of UIP and a consideration of its most common causes, including IPF (Figs. 3-16, 3-17, and 3-20) (15,64,65); collagen-vascular diseases (66), most notably rheumatoid arthritis (Fig. 3-18) (67) and scleroderma (68); drug-related fibrosis; asbestosis and other pneumoconioses (69,70); chronic HP; diffuse alveolar damage (DAD) resulting from acute respiratory distress syndrome Honeycombing Subpleural, posterior, lower lobe predominant; no atypical findings Upper-, mid-lung, or peribronchial predominance Atypical findings also present (GGO, nodules, consolidation, cysts, mosaic perfusion, air trapping) IPF (70%) Collagen-vascular disease Drug-related fibrosis Asbestosis Chronic HP Sarcoidosis (rare) Fibrotic NSIP (rare) Chronic HP Sarcoidosis Radiation Fibrotic ARDS IPF Collagen-vascular disease Drug-related fibrosis Interstitial pneumonias other than UIP (NSIP, OP, LIP, DIP) Chronic HP Sarcoidosis Cystic lung disease FIGU RE 3-20 Algorithmic approach to the differential diagnosis of honeycombing. #152201 Cust: LWW Au: Webb Pg. No. 85 85 DESIGN SERVICES OF 86 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease TABLE 3-4 Differential Diagnosis of Honeycombing Diagnosis Comments IPF Common (70%); peripheral, basal, and subpleural predominance Collagen-vascular disease Common; any collagen-vascular disease but most common in rheumatoid arthritis and scleroderma Drug-related fibrosis Many drugs possible; may be indistinguishable from other causes Asbestosis Common in advanced disease; peripheral, basal, and subpleural predominance HP (chronic) Common in advanced disease; may be peripheral, patchy, or diffuse; midlung predominance common DAD in the ARDS Honeycombing in some; may be anterior Pleuroparenchymal fibroelastosis Upper-lobe, peripheral predominance; pleural thickening Radiotherapy Localized to the radiation port Sarcoidosis A few percent of cases; may be peripheral or patchy; upper-lobe predominance common; associated with peribronchovascular fibrosis NSIP (fibrotic) Uncommon and minimal extent; other findings usually predominate Other idiopathic IPs (i.e., DIP, RB-ILD, OP, AIP, LIP) Uncommon and minimal extent; other findings usually predominate Silicosis/CWP, other pneumoconioses Uncommon (ARDS) or radiotherapy; fibrosis in some smokers (71,72); and in association with interstitial pneumonias (IPs) other than UIP (Table 3-4). For example, in a study of HRCT appearances of 129 proven cases of idiopathic IP, admittedly including atypical cases requiring biopsy for diagnosis, honeycombing was visible in 71% of patients with UIP, 39% of patients with desquamative interstitial pneumonia (DIP), 30% of patients with acute interstitial pneumonia (AIP), 26% of patients with nonspecific interstitial pneumonia (NSIP), and 13% of patients with OP (73). Honeycombing with a basal predominance was found in 59% of patients with UIP, 26% of patients with DIP, 22% of patients with NSIP, and 4% of patients with OP (73). However, honeycombing is significantly less extensive in patients with IPs other than UIP (15). In a study by Sumikawa et al. (15), the extent of honeycombing averaged 4.4% of lung parenchyma in UIP; 0.3% and 0.6% in patients with cellular and fibrotic NSIP, respectively; 0.7% in DIP or respiratory bronchiolitis- interstitial lung disease (RB-ILD); and 0.2% in LIP. In a survey of patients with end-stage lung (58), subpleural honeycombing was present in 96% of patients with UIP associated with IPF or rheumatoid arthritis, in 100% of asbestosis patients, in 44% of those with sarcoidosis, and in 75% of those with chronic HP (58,74). Honeycombing is relatively uncommon and limited in extent in patients with NSIP (75–77), but may be seen in a few percent of patients with fibrotic NSIP (65). The distribution of honeycombing is of value in differential diagnosis (Fig. 3-20). Honeycombing in patients with IPF and asbestosis is usually most severe in the subpleural lung regions and at the lung bases (63). The honeycombing in chronic HP may be most marked in the subpleural lung regions, but is more often patchy in distribution, and tends to be most severe in the midlung #152201 Cust: LWW Au: Webb Pg. No. 86 zones with relative sparing of the lung bases (58,74). Honeycombing in sarcoidosis often has an upper-lobe predominance. In patients who have pulmonary fibrosis resulting from ARDS (78), findings of fibrosis and honeycombing on follow-up HRCT had a striking anterior distribution. This distribution of reticular opacities and lung fibrosis is unusual in other diseases. Lung fibrosis limited to anterior lung regions probably reflects the fact that patients with ARDS typically develop posterior lung atelectasis and consolidation during the acute phase of their disease; it is believed that consolidation protects the posterior lung regions from the effects of mechanical ventilation, including high ventilatory pressures and high oxygen tension (78). Honeycombing in the Diagnosis of UIP and IPF In patients who present with clinical features of UIP and lack a history of collagen-vascular disease or exposure to dusts, organic antigens, or drugs, the presence of a predominantly subpleural and basal distribution of fibrosis and honeycombing on HRCT can be sufficiently characteristic of IPF to obviate biopsy (63,79–81). HRCT findings, including the presence of honeycombing with a subpleural and basal predominance, have been shown to be highly accurate in making this diagnosis (58,63,82–90). In a study by Hunninghake et al. (89) of 91 patients with idiopathic IP, clinical, physiologic, chest radiographic, and CT features were prospectively recorded; 54 patients (59%) received a pathologic diagnosis of UIP/IPF. On multivariate analysis, lower-lung honeycombing (odds ratio, 5.36) and upper-lung irregular lines (odds ratio, 6.28) were the only independent predictors of UIP/IPF. Using only these two factors, a diagnosis of UIP/IPF could be established DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities TABLE 3-5 HRCT Findings Predicting a UIP Pattern (All are Necessary) Basal and subpleural-predominant distribution Reticular opacities, traction bronchiectasis (i.e., findings supportive of fibrosis) Honeycombing Absence of inconsistent findings (see Table 3-6) with a sensitivity of 74%, a specificity of 81%, and a positive predictive value of 85%. A recent paper (63) presented the cooperative statement of the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association regarding the diagnosis of UIP and IPF. The numerous authors concluded that, in the absence of a lung biopsy, a clinical diagnosis of IPF can be solely based on the presence of a UIP pattern on HRCT and the absence of a history of diseases or exposures usually associated with this pattern (i.e., collagen disease, drugs, dusts, or organic antigens). Keep in mind that the differential diagnosis of a UIP pattern is shorter than the differential diagnosis of honeycombing, and typically includes IPF, collagen-vascular diseases, asbestosis, drug reactions, and sometimes HP. The authors, in turn, specify that the HRCT diagnosis of a UIP pattern should be based on four criteria, all of which must be present (Table 3-5, Fig. 3-21) (63). These are: 6.mosaic perfusion or air trapping, bilateral and in three or more lobes (Fig. 3-22), and 7. segmental or lobar consolidation. The presence, extent, and progression of honeycombing are important in determining the prognosis of patients with IPF and other interstitial lung diseases (91,92). A recent study (92) assessed the prognostic implications of CT and physiologic variables at baseline and on sequential evaluation in patients with fibrosing IP, both idiopathic and associated with collagen vascular disease. A 1. the presence of a basal and subpleural predominance of abnormalities, 2. reticular opacities with or without traction bronchiectasis, 3. honeycombing, and 4. the absence of findings inconsistent with the diagnosis. This combination of four findings predicts a pathologic diagnosis of UIP in 95% to 100% of cases. However, that not all cases of UIP will meet these criteria. These criteria are specific, but likely not sensitive. A HRCT diagnosis of a “possible UIP pattern” is based on the same criteria, but with honeycombing being absent. The same authors describe HRCT findings that are considered inconsistent with a UIP pattern (63). Each of these findings is typical of an IP other than UIP or a different lung disease (Table 3-6). Any one of these findings is sufficient for determining that HRCT is inconsistent with a UIP pattern. These findings include: 1. an upper- or midlung predominance of abnormalities (Fig. 3-11C), 2.a peribronchovascular predominance of abnormalities (Fig. 3-12), 3.extensive ground-glass opacity, exceeding reticulation in extent, 4. profuse micronodules, bilateral and upper lobe, 5. discrete cysts, not representing honeycombing, #152201 Cust: LWW Au: Webb Pg. No. 87 87 B C FIGU RE 3-21 A UIP pattern in a patient with IPF. A–C: Fibrosis is characterized by reticular opacities, traction bronchiectasis, and areas of honeycombing (arrows). The honeycomb cysts are seen in rows and clusters. Findings of fibrosis predominate in the bases and subpleural regions. No atypical findings are present. DESIGN SERVICES OF 88 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease TABLE 3-6 Features Inconsistent with a UIP Pattern HRCT feature Likely alternative diagnoses Ground-glass opacity outside areas of fibrosis, exceeding reticulation NSIP, HP, other IPs Mosaic perfusion/air trapping HP Centrilobular nodules HP Perilymphatic nodules Sarcoidosis, pneumoconioses Peripheral fibrosis with only minimal honeycombing, subpleural sparing NSIP Upper-lobe distribution of abnormalities Sarcoidosis, pneumoconioses, HP Parahilar, peribronchovascular predominance NSIP, HP, sarcoidosis, pneumoconioses Lower-lobe distribution of findings, not subpleural-predominant HP, NSIP The only independent predictors of mortality were the baseline extent of honeycombing and progression of honeycombing on follow-up studies (p = 0.001 and 0.002, respectively). Neither baseline nor serial change in physiologic variables, nor the presence of collagen vascular disease, was predictive of rate of survival. Variability in the Diagnosis of Honeycombing Although it may seem that honeycombing should be easily diagnosed, with good interobserver agreement, this is not always the case. Subtle or early honeycombing B A C D FIGU RE 3-22 HRCT inconsistent with UIP and IPF in a patient with chronic HP and honeycombing. A and B: Inspiratory images show reticulation, traction bronchiectasis, and honeycombing (arrows) in the subpleural lung. C and D: Dynamic expiratory images at the same levels show air trapping in multiple lobules (arrows) in three lobes, a finding inconsistent with UIP. #152201 Cust: LWW Au: Webb Pg. No. 88 DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities may be difficult to distinguish from pure reticulation or traction bronchiectasis, and other abnormalities such as paraseptal emphysema and cystic lung diseases may be confused with this finding (93). In a recent study (93) assessing interobserver variability in the diagnosis of honeycombing, five experts scored 80 HRCT images for the presence of honeycombing using a 5-point scale (5 = definitely present to 1 = definitely absent) to establish a reference standard. Forty-three observers, a number of whom were expert thoracic radiologists, subsequently scored the HRCT using the same scoring system. Their agreement with the reference standard was only moderate (weighted k values: 0.40–0.58); of note, agreement among the five study experts as to the presence of honeycombing was only slightly better (k value = 0.45– 0.67). On a case-by-case basis, observers agreed that honeycombing was present in 21 of the 80 (26%) cases and agreed that honeycombing was absent in 18 (22%). They disagreed as to the presence of honeycombing in 23 images (29%); these cases included honeycombing mixed with traction bronchiectasis, large cysts, and fibrosis with superimposed emphysema. The remaining 18 images (22%) did not fulfill the criteria of the previous three categories. 89 Intralobular Interstitial Thickening (Intralobular Lines) Intralobular interstitial thickening results in a fine reticular pattern on HRCT, with the visible lines separated by a few millimeters (Fig. 3-1) (52). Lung regions showing this finding characteristically show a fine lace- or netlike appearance (Figs. 3-1 and 3-23 to 3-28). Intralobular interstitial thickening is a nonspecific finding; it may be associated with interstitial fibrosis (Figs. 3-23 to 3-26) or interstitial infiltration or inflammation in the absence of fibrosis (Figs. 3-27 and 3-28). The presence of intralobular interstitial thickening is described using the term intralobular lines (10,52). This finding is responsible for the “small reticular pattern” originally described by Zerhouni et al. (12). Intralobular lines may be seen in isolated or associated with interlobular septal thickening or honeycombing. In patients with intralobular interstitial thickening resulting from fibrosis, intralobular bronchioles may be visible in the peripheral lung (Figs. 3-1 and 3-23). This is not a normal finding, and it results from a combination of bronchiolar dilatation (i.e., traction bronchiolectasis) and thickening of the peribronchiolar interstitium that A B FIGU RE 3-23 Intralobular interstitial thickening and traction bronchiolectasis in a patient with IPF. A: Prone HCRT shows a fine network of lines in the lung periphery. Intralobular bronchioles (arrows) are visible throughout the peripheral lung as a result of fibrosis and traction bronchiolectasis. B: Histologic specimen in a patient with IPF shows fibrosis, intralobular interstitial thickening, and bronchiolecstasis (br.) (Courtesy of Martha Warnock, MD.) #152201 Cust: LWW Au: Webb Pg. No. 89 DESIGN SERVICES OF 90 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease FIGU RE 3-24 Intralobular interstitial thickening in a patient with early IPF. On a supine scan, fine reticular opacities are visible posteriorly. This abnormality reflects intralobular interstitial thickening. A surrounds them (52,57). Traction bronchiectasis, dilatation of large bronchi occurring because of fibrosis, may also be seen (Figs. 3-25 and 3-26). Traction bronchiectasis and traction bronchiolectasis are described in more detail below. Intralobular interstitial thickening as perceived on HRCT reflects thickening of the distal peribronchovascular B A C FIGURE 3-26 Prone scans in a patient with IPF. A: Abnormal reticulation representing intralobular interstitial thickening predominates in the subpleural lung. B: At a lower level, fibrosis is more extensive. Traction bronchiectasis and bronchiolectasis are predominant features. Also, note the irregular thickening of the major fissure (large arrow) and irregular interlobular septal thickening. C: Typically, traction bronchiectasis and bronchiolectasis are characterized by irregular, varicose, or cockscrew appearance (arrows). B FIGU RE 3-25 Prone scans in a patient with fibrotic NSIP. A and B: Abnormal reticulation represents intralobular interstitial thickening. Traction bronchiectasis (arrows) is easily seen. #152201 Cust: LWW Au: Webb Pg. No. 90 interstitial tissues and the intralobular interstitium. As an isolated finding, it is most commonly seen in patients with pulmonary fibrosis (Figs. 3-23 to 3-27). In patients who have IPF or other causes of UIP, such as rheumatoid arthritis, scleroderma, or other collagen-vascular DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 91 FIGU RE 3-27 Cellular NSIP with intralobular interstitial thickening and ground-glass opacity. Ground-glass opacity predominates in the posterior lung, with subpleural sparing typical of NSIP. Fine reticular opacities in association with the ground-glass represent intralobular lines. diseases, fibrosis tends to predominantly involve alveoli in the periphery of acini, resulting in a “peripheral acinar distribution” of interstitial fibrosis (30,57); this histologic finding correlates with the presence of intralobular lines on HRCT. Intralobular lines, resulting in a fine reticular p attern, can also be seen in patients with NSIP or other IPs (Table 3-7) (Figs. 3-25, 3-27, and 3-29) (15,73,75-77,94). In NSIP, the presence of intralobular lines or irregular linear opacities correlated with the presence of interstitial fibrosis and was often associated with bronchial or bronchiolar dilatation (traction bronchiectasis or bronchiolectasis) (75,95). In a study of HRCT appearances of various idiopathic IPs, intralobular lines were visible in 97% of patients with UIP, 93% of patients with NSIP, 78% of patients with DIP, 71% of patients with OP, and F I G U R E 3-28 Pulmonary hemorrhage with intralobular interstitial thickening and ground-glass opacity. Patchy ground-glass opacity and intralobular lines represent focal pulmonary hemorrhage. 70% of patients with AIP (73). Intralobular lines are also common in asbestosis (96). Intralobular interstitial thickening can be seen in the absence of significant fibrosis in patients with a variety of infiltrative or inflammatory lung diseases (Table 3-7) (Figs. 3-27 to 3-29) (97). When this is the case, traction bronchiectasis and other manifestations of fibrosis are less evident or absent, and ground-glass opacity may be visible instead. Intralobular interstitial thickening may be seen in association with interlobular septal thickening in patients with diseases such as pulmonary edema, TABLE 3-7 Differential Diagnosis of Intralobular Interstitial Thickening Diagnosis Comments IPF or other cause of UIP Common (97%); often associated with honeycombing HP (chronic) Common; associated with other findings of fibrosis Asbestosis Common; associated with other findings of fibrosis NSIP Common (93%); ground-glass opacity (cellular NSIP) or traction bronchiolectasis (fibrotic NSIP) commonly visible Other idiopathic IPs (i.e., DIP, OP, AIP) Common (70%); other findings (i.e., traction bronchiolectasis, ground-glass opacity, consolidation also present) Lymphangitic carcinomatosis, lymphoma, leukemia Smooth or nodular; associated with septal thickening Pulmonary edema Smooth; associated with septal thickening and ground-glass opacity Pulmonary hemorrhage Smooth; associated with septal thickening and ground-glass opacity Pneumonia (e.g., viral, Pneumocystis carinii) Smooth; associated with septal thickening and ground-glass opacity Alveolar proteinosis Smooth; associated with septal thickening and ground-glass opacity Other causes of septal thickening, lung fibrosis, or lung infiltration See interlobular septal thickening, honeycombing, crazy paving differential diagnoses #152201 Cust: LWW Au: Webb Pg. No. 91 DESIGN SERVICES OF 92 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease Intralobular lines Traction bronchiectasis also present Septal thickening also present Ground-glass opacity also present Fibrosis Septal thickening differential diagnosis Infiltrative or inflammatory disease Consider causes of honeycombing and fibrotic lung disease “Crazy paving” differential diagnosis; e.g., edema Hemorrhage Atypical infections Interstitial pneumonias HP Adenocarcinoma FIGU RE 3-29 Algorithmic approach to the differential diagnosis of intralobular lines. pulmonary hemorrhage, atypical pneumonia, lymphangitic spread of carcinoma, and other infiltrative processes such as cellular NSIP (7). In the absence of traction bronchiectasis or bronchiolectasis, the differential diagnosis of this finding is identical to that of interlobular septal thickening. Intralobular lines may also be seen in patients with ground-glass opacity or the pattern of crazy paving, in association with diseases such as pulmonary edema or hemorrhage (25), some pneumonias (e.g., Pneumocystis jiroveci, cytomegalovirus), IPs such as NSIP and OP, HP, invasive mucinous adenocarcinoma, and alveolar proteinosis (Figs. 3-8 and 3-29). NONSPECIFIC RETICULATION Generally speaking, if a reticular pattern visible on HRCT cannot be characterized as representing interlobular septal thickening, honeycombing, or intralobular interstitial thickening (intralobular lines), the nonspecific terms “reticulation,” “reticular pattern,” or “reticular opacities” can be used to describe the abnormality present (10). Reticular opacities 1 to 3 mm thick, that cannot be characterized as representing one of these patterns, are often visible in patients with interstitial disease, representing bands of fibrosis or other causes of interstitial infiltration (9). Such opacities may represent poorly characterized irregular interlobular septal thickening, poorly characterized areas of honeycombing, bands of fibrous tissue in the periphery of pulmonary lobules or bridging the lobule from the centrilobular region to the lobular periphery, or thickening of the intralobular interstitium by fibrous tissue or because of inflammation or infiltration. #152201 Cust: LWW Au: Webb Pg. No. 92 This finding is nonspecific and may be seen in a v ariety of diseases, including those associated with inflammation or fibrosis, including UIP and NSIP (73,75–77), and in a percentage of cigarette smokers (71,72). In patients who have UIP, irregular reticulation may be seen instead of honeycombing, particularly in early cases or in less abnormal regions (i.e., in the upper lobes of patients with IPF); in patients with NSIP, reticulation is more common than honeycombing. THE INTERFACE SIGN The presence of irregular interfaces between the aerated lung parenchyma and bronchi, vessels, or visceral pleural surfaces has been termed the interface sign by Zerhouni et al. (1,12) (Figs. 3-1 and 3-30). The interface sign is nonspecific and is commonly seen in patients with an interstitial abnormality, regardless of its cause. In the original description of the interface sign, this finding was visible in 89% of patients with interstitial lung disease (12). The interface sign is generally associated with an increase in lung reticulation; the presence of thin linear opacities contacting the bronchi, vessels, or pleural surfaces is responsible for their having an irregular or spiculated appearance on HRCT (Fig. 3-30). The linear opacities producing the interface sign may represent thickened interlobular septa, intralobular lines, or irregular scars (Fig. 3-1). The interface sign is most frequently visible in patients with fibrotic lung disease, but it may also be seen in patients with infiltrative diseases, inflammatory disease, and pulmonary edema. Nishimura et al. (57) reported the presence of irregular pleural surfaces and irregular vessel margins in 94% and 98%, respectively, of patients with IPF. In virtually all cases showing DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities FIGU RE 3-30 The interface sign in fibrotic sarcoidosis. The irregular appearance of the fissure and the edges of vessels reflects the presence of increased lung reticulation, whether due to intralobular lines, septal thickening, or honeycombing. the interface sign, other, more specific, abnormal findings will also be visible on HRCT. TRACTION BRONCHIECTASIS AND TRACTION BRONCHIOLECTASIS In patients with lung fibrosis and a reticular pattern visible on HRCT, bronchial dilatation is commonly present, resulting from traction by fibrous tissue on the bronchi walls. This is termed traction bronchiectasis (Figs. 3-1 and 3-31); it typically results in irregular bronchial dilatation with the abnormal bronchus appearing varicose or “corkscrew” (52,98). Traction bronchiectasis usually involves the segmental and subsegmental bronchi and is most commonly visible in the perihilar regions in patients with significant lung fibrosis (Figs. 3-25 and 3-26) (49,99). It can also affect small peripheral bronchi or bronchioles (Fig. 3-23), an occurrence termed traction bronchiolectasis. The branching or irregular tubular appearance of traction bronchiectasis or bronchiolectasis should be distinguished from honeycombing whenever possible. As reviewed above, the diagnosis of honeycombing should be limited to cases showing rows, layers, or clusters of thick-walled air cysts in the immediate subpleural lung. In a patient with fibrosis, thick-walled, air-filled cysts separated from the pleural surface often represent traction bronchiectasis or traction bronchiolectasis. The presence of traction bronchiectasis usually indicates that lung fibrosis is present, and the differential diagnosis includes a large number of fibrotic lung diseases. Common lung diseases associated with fibrosis and traction bronchiectasis, but without visible honeycombing, #152201 Cust: LWW Au: Webb Pg. No. 93 93 FIGU RE 3-31 Traction bronchiectasis and traction bronchiolectasis in pulmonary fibrosis. A thin lung slice from a patient with IPF shows honeycombing in the posterior lung. Irregular dilatation of bronchi (traction bronchiectasis) having a varicose or corkscrew appearance (red arrows) reflects the presence of lung fibrosis. Dilatation of bronchioles (traction bronchiolectasis, blue arrows) in less-abnormal lung regions also reflects surrounding lung fibrosis. include fibrotic NSIP, end-stage sarcoidosis, and chronic HP, but keep in mind that UIP may also result in lung fibrosis without honeycombing. When honeycombing is also present, UIP is most likely. Some patients with cellular NSIP and other inflammatory lung diseases can show bronchial dilatation resembling traction bronchiectasis that resolves with the resolution of the lung disease. Often, bronchial dilatation in these patients is cylindrical rather than irregular and varicose, and findings of inflammation such as ground-glass opacity or consolidation predominate in the abnormal regions. Likely, a decrease in lung compliance associated with interstitial infiltration results in bronchial dilatation in these cases. In patients with lung fibrosis and intralobular interstitial thickening, intralobular bronchioles may be visible because of their dilatation (traction bronchiolectasis) and surrounding fibrous tissue (Fig. 3-23). The differential diagnosis of this finding is that of lung fibrosis. Akira et al. (69) found traction bronchiolectasis to be more common in patients with IPF (78%) than in those with asbestosis (20%). In some cases, the HRCT pattern of intralobular lines can reflect the presence of very small honeycomb cysts or dilated bronchioles associated with surrounding lung fibrosis. Nishimura et al. (57) reviewed 46 cases of IPF with UIP, correlating findings on CT with appearances of lung histology from open biopsy specimens or autopsy. Visibility of centrilobular bronchioles in association with a fine reticulation or increased lung attenuation was found in 96% of cases, indicating the DESIGN SERVICES OF 94 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease presence of bronchiolar dilatation, fibrosis, and “microscopic” honeycombing, with dilated bronchioles or small cysts measuring approximately 1 mm in diameter (57). PERIBRONCHOVASCULAR INTERSTITIAL THICKENING Central bronchi and pulmonary arteries are surrounded and enveloped by a strong connective tissue sheath, termed the peribronchovascular interstitium, extending from the level of the pulmonary hila into the peripheral lung. In the lung periphery, the peribronchovascular interstitium surrounds centrilobular arteries and respiratory bronchioles (Fig. 2-1) (100). The peribronchovascular interstitium has been termed the axial interstitium by Weibel (101). Thickening of the perihilar peribronchovascular interstitium occurs in many diseases that cause a generalized interstitial abnormality (Table 3-8) (2,4,7,102). Peribronchovascular interstitial thickening is common in patients with lymphangitic spread of carcinoma (4,7,103); lymphoma (104); leukemia (105); lymphoproliferative disease such as LIP (43–45); interstitial pulmonary edema (23,106); diseases that result in a perilymphatic distribution of nodules (e.g., sarcoidosis) (49); and in many diseases that result in pulmonary fibrosis, particularly sarcoidosis, which has a propensity to involve the peribronchovascular interstitium (48,107). Peribronchovascular interstitial thickening has also been reported in as many as 65% of patients with NSIP (75) and 19% of patients with chronic HP (74). Because the thickened peribronchovascular interstitium cannot be distinguished from the underlying opacity of the bronchial wall or pulmonary artery, this abnormality is usually perceived on HRCT as an increase in bronchial wall thickness or an increase in diameter of pulmonary artery branches (Fig. 3-32) (7). Apparent bronchial wall thickening is the easier of these two findings to recognize. This finding is exactly equivalent to “peribronchial cuffing” seen on plain chest radiographs in patients with an interstitial abnormality. In patients with pulmonary interstitial emphysema, air is commonly seen within the peribronchovascular interstitium, outlining vessels and bronchi (Fig. 3-32D) (108–110). Peribronchovascular interstitial thickening is commonly present in patients with interlobular septal thickening, and as with septal thickening, it can appear smooth, nodular, or irregular in different diseases (Figs. 3-2 to 3-5) (100). Smooth peribronchovascular interstitial thickening is most typical of patients with lymphangitic spread of carcinoma or lymphoma (Fig. 3-33) and interstitial pulmonary edema (23,106), but can be seen in patients with fibrotic lung disease as well. Nodular thickening of the peribronchovascular interstitium is particularly common in sarcoidosis (Fig. 3-34) and lymphangitic spread of carcinoma. The presence of irregular peribronchovascular interstitial thickening, as an example of the interface sign, is most frequently seen in patients with adjacent lung fibrosis. Extensive peribronchovascular fibrosis can also result in the presence of large conglomerate masses of fibrous tissue (Fig. 3-35). This can occur in patients with sarcoidosis, silicosis, tuberculosis, and talcosis, (49,107,111) and is discussed in greater detail in Chapter 4. Peribronchovascular interstitial thickening is easy to diagnose if it is marked; in this instance, bronchial walls appear several millimeters thick, and bronchovascular structures may show evidence of the interface sign or nodules. However, the diagnosis of minimal peribronchovascular thickening can be difficult and is quite subjective, particularly if the abnormality is diffuse and symmetric. Although the thickness of the wall of a normal bronchus should measure from one-sixth to one-tenth of its diameter (see Chapter 2) (112), and in several HRCT studies, a veraged about 0.2 of the bronchial diameter (113,114), there are no reliable criteria as to what represents the upper limit of normal for the combined thickness of bronchial wall and the surrounding interstitium (115). Furthermore, these measurements vary, depending on the lung window chosen, and too low a window mean can make normal bronchi or vessels appear abnormal. However, in many patients with peribronchovascular interstitial thickening, and particularly in patients with lymphangitic spread of carcinoma TABLE 3-8 Differential Diagnosis of Peribronchovascular Interstitial Thickening Diagnosis Comments Lymphangitic carcinomatosis, lymphoma, leukemia Common; smooth or nodular; may be the only abnormality Lymphoproliferative disease (e.g., LIP) Smooth or nodular; other abnormalities typically present Pulmonary edema Common; smooth Sarcoidosis Common; usually nodular or irregular; conglomerate masses of fibrous tissue with traction bronchiectasis typical in end stage IPF or other cause of UIP Common; often irregular; associated with traction bronchiectasis; other findings of fibrosis predominate NSIP With findings of ground-glass opacity and reticulation Silicosis/CWP, talcosis Conglomerate masses HP (chronic) Sometimes visible; often irregular; associated with traction bronchiectasis #152201 Cust: LWW Au: Webb Pg. No. 94 DESIGN SERVICES OF Paired bronchi and arteries of similar size (bronchoarterial ratio < 1) Smooth Nodular Irregular Peribronchovascular interstitial thickening Normal Signet ring sign Bronchiectasis FIGURE 3-32 Differentiation of peribronchovascular interstitial thickening and bronchiectasis. A: In a normal subject, bronchi are uniformly thin-walled and appear approximately equal in diameter to adjacent pulmonary arteries. B: In the presence of peribronchovascular interstitial thickening, there appears to be an increase in bronchial wall thickness and a corresponding increase in the diameter of pulmonary artery branches. The contours of the bronchi and vessels can appear smooth, nodular, or irregular in different diseases. C: In bronchiectasis, bronchi are usually thick walled and appear larger than adjacent pulmonary arteries. This results in the so-called signet ring sign. D: CT with 3-mm collimation in a patient with pulmonary interstitial emphysema. Air is visible within the peribronchovascular interstitial sheath, outlining pulmonary arteries (large black arrows) and bronchi (small black arrow). Air also surrounds pulmonary veins. FIGU RE 3-33 Peribronchovascular interstitial thickening. In a patient with unilateral lymphangitic spread of carcinoma including the left lung, there is smooth thickening of the peribronchovascular interstitium manifested as peribronchial cuffing (arrows); this appearance is easily contrasted with that of normal bronchi in the right lung. Note that the left-sided pulmonary artery branches appear similar in diameter to the cuffed bronchi because the thickened interstitium surrounds them as well. Small intrapulmonary vessels on the left also appear more prominent than those on the normal side because of perivascular interstitial thickening. Interlobular septal thickening and subpleural nodules are also visible on the left. 95 #152201 Cust: LWW Au: Webb Pg. No. 95 DESIGN SERVICES OF 96 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease and confusion between these two is not often a problem in clinical practice. In addition, several HRCT findings allow these two entities to be differentiated (Fig. 3-32). First, peribronchovascular interstitial thickening is often associated with other findings of interstitial disease, such as septal thickening, irregular reticulation, intralobular lines, honeycombing, or the interface sign, whereas bronchiectasis usually is not. Second, in patients with bronchiectasis, the abnormal thick-walled and dilated bronchi often appear much larger than the adjacent pulmonary artery branches, with a bronchoarterial ratio exceeding 1 (see Chapter 2) (Fig. 3-36). This results in the appearance of large ring shadows, each associated FIGU RE 3-34 Nodular peribronchovascular interstitial thickening in a patient with sarcoidosis. Numerous small nodules surround the central bronchi and vessels. and sarcoidosis, this abnormality is unilateral or patchy, sparing some areas of lung. In such patients, normal and abnormal lung regions can easily be contrasted (Fig. 3-33). As a rule, bronchial walls in corresponding regions of one or both lungs should be similar in thickness. Bronchial wall thickening occurring in patients with true bronchiectasis or chronic obstructive pulmonary disease (COPD) produces an abnormality that closely mimics the HRCT appearance of peribronchovascular interstitial thickening. However, airway diseases and interstitial diseases can usually be distinguished on the basis of symptoms or pulmonary function abnormalities, A B FIGU RE 3-35 Peribronchovascular interstitial thickening in end-stage sarcoidosis, with conglomerate masses of fibrous tissue surrounding the central vessels and bronchi. Bronchi appear dilated and thick walled because of surrounding fibrosis and traction bronchiectasis. Note that the vessels and bronchi appear to be of similar diameter. #152201 Cust: LWW Au: Webb Pg. No. 96 FIGU RE 3-36 Bronchiectasis with the signet ring sign. A: Thick-walled and dilated bronchi (large arrows) appear larger than the adjacent pulmonary artery branches (small arrows). This appearance is termed the signet ring sign and is typical of bronchiectasis. B: In another patient, upper-lobe bronchiectasis is present, with several good examples of the signet ring sign (arrows). The signet ring sign indicates that the bronchoarterial ratio is abnormally increased. DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities with a small, rounded opacity, a finding that has been termed the signet ring sign, and is considered to be diagnostic of bronchiectasis (116–120). In patients with peribronchovascular interstitial thickening, however, the size relationship between the bronchus and artery is maintained, and they appear to be of approximately equal size. The diagnosis and appearances of bronchiectasis and bronchial wall thickening is discussed in greater detail in Chapter 6. Diseases that cause peribronchovascular interstitial thickening often result in prominence of the centrilobular artery, which normally appears as a dot, Y-shaped, or X-shaped branching opacity. This finding reflects thickening of the intralobular component of the peribronchovascular interstitium, also termed the cen trilobular interstitium (Fig. 2-1) (2,4,8,12). On HRCT, linear, branching, or dotlike centrilobular opacity may be seen (Fig. 3-1). Thickening of the centrilobular interstitium is often associated with interlobular septal thickening or intralobular interstitial thickening (Fig. 3-1) but sometimes occurs as an isolated abnormality. Centrilobular interstitial thickening is common in patients with lymphangitic spread of carcinoma or lymphoma (4,7) and interstitial pulmonary edema (23,121). In patients with lung fibrosis, centrilobular interstitial thickening is common but almost always associated with honeycombing, traction bronchiolectasis, or intralobular lines. However, it is significantly more extensive in patients with NSIP than UIP (15). PARENCHYMAL BANDS The term parenchymal band has been used to describe a nontapering, reticular opacity, usually 1 to 3 mm in thickness and up to 5 cm in length, seen in patients with atelectasis, pulmonary fibrosis, or other causes of interstitial thickening, and often associated with pleural thickening (Figs. 3-1, 3-36, and 3-37) (8–10,122). A parenchymal band is often peripheral and generally contacts the visceral pleural surface, which may be thickened and retracted inward. In some patients, these bands represent contiguous thickened interlobular septa and have the same significance and differential diagnosis as septal thickening (11). When parenchymal bands can be identified as thickened septa, the use of a separate term to describe this finding is unjustified; the term septal thickening should suffice. However, parenchymal bands visible on HRCT can also represent areas of peribronchovascular fibrosis, coarse scars, or atelectasis associated with lung infiltration or pleural fibrosis (Figs. 3-37 and 3-38) (11,123). These nonseptal bands are often several millimeters thick and irregular in contour and are associated with significant distortion of adjacent lung parenchyma and bronchovascular structures (124). Parenchymal bands may be seen in various diseases, and have been reported as most common in patients #152201 Cust: LWW Au: Webb Pg. No. 97 97 FIGU RE 3-37 Parenchymal bands in a patient with asbestos. A prone scan shows both thick and thin bands. These may be seen in relation to visceral pleural thickening, representing scars or atelecstasis. with asbestos-related lung and pleural disease (Fig. 3-37), sarcoidosis with interstitial fibrosis (49), silicosis associated with progressive massive fibrosis and conglomerate masses, tuberculosis, and lung disease associated with ankylosing spondylitis (53,125–127) (Table 3-9). In patients with asbestos exposure, multiple parenchymal bands are common; in one study (8), multiple parenchymal FIGU RE 3-38 Parenchymal band in a patient with otherwise normal lungs. This likely represents an isolated scar. DESIGN SERVICES OF 98 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease TABLE 3-9 Differential Diagnosis of Parenchymal Bands Diagnosis Comments Asbestosis Multiple parenchymal bands common; smooth; associated with thickened pleura Sarcoidosis Common; associated with septal thickening Silicosis/CWP In association with progressive massive fibrosis and emphysema Tuberculosis Associated with scarring IPF or other cause of UIP Common; often irregular; associated with traction bronchiectasis; other findings of fibrosis predominate NSIP With findings of ground-glass opacity and reticulation Silicosis/CWP, talcosis Conglomerate masses HP (chronic) Sometimes visible; often irregular; associated with traction bronchiectasis Ankylosing spondylitis Apical bands were seen in 66% of asbestos-exposed patients. In patients with asbestos-related disease, parenchymal bands can reflect thickened interlobular septa, indicating pulmonary fibrosis, or, more often, areas of atelectasis and focal scarring occurring in association with visceral pleural thickening or pleural plaques. In asbestos-exposed patients, parenchymal bands are frequently associated with areas of thickened pleura and have a basal predominance (8,123). They may precede the development of rounded atelectasis. SUBPLEURAL INTERSTITIAL THICKENING Usually, thickening of the interlobular septa within the peripheral lung is associated with thickening of the subpleural interstitium (1,2); both the septa and the subpleural interstitium are part of the peripheral interstitial fiber system described by Weibel (Fig. 2-1) (101). Subpleural interstitial thickening can be difficult to recognize in locations where the lung contacts the chest wall or mediastinum but is easy to see adjacent to the major fissures (Figs. 3-1 to 3-3). Because two layers of the subpleural interstitium are seen adjacent to each other in this location, any subpleural abnormality appears twice as abnormal as it does elsewhere. Thus, thickening of the fissure visible on HRCT often represents subpleural interstitial thickening. If the thickening is smooth, it may be difficult to distinguish from fissural fluid. If the interface sign is present and the thickening is irregular in appearance (1,12), or if the thickening is nodular, an interstitial abnormality is more easily diagnosed. In general, the differential diagnosis of subpleural interstitial thickening is the same as that of interlobular septal thickening, although subpleural interstitial thickening is more common than septal thickening in patients with IPF or UIP of any cause. The presence of subpleural interstitial fibrosis with irregular or “rugged” pleural surfaces has been reported by Nishimura et al. (57) as a common finding in IPF, correlating with the presence of fibrosis predominantly affecting the lobular periphery; #152201 Cust: LWW Au: Webb Pg. No. 98 this finding was present in 94% of the cases of IPF that he studied. A subpleural predominance of fibrosis can also be seen in patients with collagen-vascular diseases and drug-related fibrosis (30). Nodular thickening of the subpleural interstitium can also be seen, and it has the same differential diagnosis as nodular septal thickening (47). Remy-Jardin et al. (47) reported the appearance of subpleural micronodules, defined as 7 mm or less in diameter, on HRCT in patients with sarcoidosis, CWP, lymphangitic spread of carcinoma, and LIP, and in a small percentage of normal subjects. Subpleural nodules are described further in the next chapter. SUBPLEURAL CURVILINEAR LINE A curvilinear opacity a few millimeters or less in t hickness, less than 1 cm from the pleural surface and paralleling the pleura, is termed a subpleural line or subpleural curvilinear line (9,10). It is a nonspecific indicator of atelectasis, fibrosis, inflammation, or even edema. It was first described in patients with asbestosis (128). It was originally suggested that a subpleural line reflected the presence of fibrosis associated with honeycombing (128), and in some patients, a confluence of honeycomb cysts can result in a somewhat irregular subpleural line (Figs. 3-39 to 3-42). A subpleural line representing fibrosis is much more common in patients who have asbestosis than in those who have IPF or other causes of UIP (54,83). Indeed, the presence of a subpleural line in nondependent lung has been reported in as many as 41% of patients with clinical findings of asbestosis (8). However, the presence of this finding is nonspecific and can be seen in a variety of lung diseases (Fig. 3-1). The presence of subpleural lines has also been reported as common in patients with scleroderma who have interstitial disease (Fig. 3-41) (129,130); this may reflect the common occurrence of NSIP in patients with scleroderma and the tendency of NSIP to spare the immediate subpleural lung (Fig. 3-42). Subpleural lines may also be seen in ankylosing spondylitis (53). DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 99 A FIGU RE 3-39 Subpleural line in a patient with asbestosis. An illdefined subpleural line (arrows) on a prone scan reflects subpleural fibrosis and honeycombing. Other findings of pulmonary fibrosis are also present. A subpleural line also has been reported to occur as a result of the confluence of peribronchiolar interstitial abnormalities in patients with asbestosis, representing early fibrosis with associated alveolar flattening and collapse (11,96). In these patients, honeycombing was not present. Also, in patients with asbestos exposure, a subpleural line may be seen adjacent to focal pleural thickening or plaques. These most likely represent focal atelectasis or localized areas of scarring. It is common to see a subpleural line or focal areas of reticulation adjacent to thoracic spine osteophytes or other causes of chest wall distortion, such as prominent healed rib fractures or at the site of postsurgical lung hernia. The subpleural line or reticulation reflects the presence of localized alveolar collapse and fibrosis (131). In addition, in some patients with asbestos exposure, a subpleural line may be seen adjacent to pleural plaques, representing focal atelectasis. A subpleural line can also be seen in normal patients as a result of atelectasis within the dependent lung (e.g., the posterior lung when the patient is positioned supine); the presence of dependent atelectasis has been confirmed experimentally (132). Also, a thicker, less well-defined B C FIGU RE 3-40 Bilateral subpleural lines (arrows) in a patient with early IPF. #152201 Cust: LWW Au: Webb Pg. No. 99 FIGU RE 3-41 Subpleural lines in a patient with scleroderma who likely has NSIP. Supine (A) and prone (B). HRCT shows bilateral subpleural lines (arrows). C: Sagittal reconstruction also shows a posterior subpleural line. DESIGN SERVICES OF 100 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease DISTRIBUTION OF LINEAR AND RETICULAR OPACITIES IN THE DIAGNOSIS OF LUNG DISEASE FIGU RE 3-42 Subpleural line in a patient with NSIP. On an axial HRCT obtained in the prone position, bilateral subpleural lines are visible. In this patient, this may reflect the tendency of NSIP to spare the immediate subpleural lung. When attempting to reach a diagnosis or differential diagnosis of lung disease using HRCT, the overall distribution of pulmonary abnormalities should be considered along with their morphology, HRCT appearance, and distribution relative to lobular structures (3,59,84,135). Many lung diseases show specific regional distributions or preferences, a fact that is likely related to their underlying pathogenesis and pathophysiology (136). An important caveat to keep in mind when reading the following section is that significant variations in classical patterns of lung involvement can be seen in individual patients. A specific diagnosis should not be excluded because of an atypical distribution of abnormalities. Central Lung Versus Peripheral Lung subpleural opacity, a so-called dependent density (8) or dependent opacity (133) can also be seen in normal subjects as a result of volume loss. In a study by Lee et al. (133), dependent lung opacity was significantly more common at reduced lung volumes, with studies using spirometrically gated HRCT. Such normal posterior lines or opacities are transient and disappear in the prone position. In a study of patients with asbestos exposure by Aberle et al. (8), neither transient subpleural lines nor transient dependent densities correlated with the clinical suspicion of pulmonary fibrosis. In patients with early interstitial lung disease, there may be a greater tendency for atelectasis to develop in the peripheral lung, resulting in the appearance of a subpleural line. As such, the presence of this abnormality could reflect an increased closing volume (i.e., an increased tendency for the lung to collapse) that is known to occur as a result of early interstitial lung disease. The association of platelike atelectasis at the junction of “cortical” and “medullary” lung regions, air trapping in the lung peripheral to the atelectasis, and decreased compliance of lung because of interstitial infiltration was first reported by Kubota et al. (134). Some diseases have a central, perihilar, bronchocentric, or peribronchovascular distribution (59,100), whereas others favor the peripheral or subpleural parenchyma, or lung cortex (Table 3-10). Diseases associated with reticular opacities on HRCT that can have a central or perihilar predominance include end-stage sarcoidosis, silicosis, fibrotic NSIP, chronic HP (137), and lymphangitic spread of carcinoma (138). In a study by Grenier et al. (138), a predominantly central distribution of abnormalities was visible in 16% of patients with sarcoidosis, 31% of patients with silicosis, and 8% of those with lymphangitic spread of carcinoma. In another study (84), a central or peribronchovascular predominance was seen in 70% of patients with sarcoidosis and 60% of patients with lymphangitic spread of carcinoma. A predominantly peribronchial distribution of fibrosis is considered inconsistent with UIP and IPF (63); in patients TABLE 3-10 Predominance of Lung Disease on HRCT: Central Lung Versus Peripheral Lung Lung disease Findings Central lung NORMAL RETICULAR OPACITIES IN THE ELDERLY Sarcoidosis Peribronchovascular nodules; conglomerate fibrosis with traction bronchiectasis Silicosis Conglomerate masses of fibrosis The presence of fine reticular opacities in the peripheral, posterior, lower lobes can be seen as a normal finding in older subjects. In a study by Copley et al. (17), a limited subpleural reticular pattern was identified in the majority (24 of 40, 60%) of individuals older than 75 years, and was absent in a group younger than 55 years (p < 0.001) (17). The reticulation was fine or coarse, and was unassociated with honeycombing or traction bronchiectasis. Interlobular septal thickening was also seen in some patients. These findings are presumably due to an increase in lung collagen in older patients. Talcosis Conglomerate masses of fibrosis Lymphangitic spread of carcinoma Peribronchovascular interstitial thickening or nodules HP Peribronchovascular fibrosis in some NSIP Peribronchovascular fibrosis in some #152201 Cust: LWW Au: Webb Pg. No. 100 Peripheral lung UIP, IPF, collagen diseases, asbestosis Subpleural fibrosis; honeycombing NSIP in 50% Subpleural ground-glass opacity; reticulation; subpleural sparing DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities with HRCT findings of fibrosis, this distribution would be more typical of NSIP (95), chronic HP, or sarcoidosis. A peripheral, cortical, or subpleural predominance of abnormalities is typical of UIP and has been reported in nearly all patients with asbestosis (58); 81% to 94% of patients with IPF (58,84,85); and a similar high percentage of patients with scleroderma, rheumatoid lung disease, or other collagen-vascular diseases. Other fibrotic IPs, such as NSIP, commonly show a peripheral predominance, although subpleural sparing is present in about half. A peripheral predominance of abnormalities is visible in approximately half of patients with OP and DIP (58,84,138–141), although these entities uncommonly present with a predominantly reticular pattern. Peripheral predominance is occasionally present in patients with chronic HP and end-stage sarcoidosis, ranging from a few percent to 20% in different studies. Subpleural predominance is also typical of diffuse amyloidosis, although this disease is quite rare. In a study by Silva et al. (137) of HRCT findings in patients with IPF, NSIP, and chronic HP with fibrosis, patients with IPF and NSIP were more likely to have peripheral predominance of abnormalities (78% and 72%, respectively) (p< 0.001) and fibrosis (100% and 92%, respectively) than those with chronic HP (25% and 78%, respectively) (p< 0.002). Upper Lung Versus Lower Lung The relative extent and severity of abnormalities in the upper lungs and midlungs and at the lung bases can be determined on HRCT by using coronal or sagittal reconstruction of volumetric HRCT, or by comparing the severity of abnormalities on transaxial scans at different levels. Some diseases tend to predominate in the upper lobes, whereas others predominate in the mid- or lower lobes (Table 3-11) (142). Diseases that can result in reticular opacities and have been recognized to have upper-lobe predominance on HRCT include sarcoidosis, silicosis, and CWP chronic HP (48,50,84,85,135,138,143–145), and Langerhans cell histiocytosis, in the small percentage cases presenting with reticulation. An upper-lobe predominance of abnormalities is present in nearly equal percentages of patients with sarcoidosis (47%–50%) and silicosis (55%–69%), whereas a lower-lobe predominance is present in less than 10% of patients with these diseases (85,138). According to recent criteria, a predominantly upper- or midlung predominance of fibrosis is considered inconsistent with a diagnosis of UIP and IPF (63); however, some patients with a diagnosis of UIP may show this distribution. In patients with HRCT findings of fibrosis, an upper- or midlung predominance would be more typical of chronic HP or sarcoidosis. A basal distribution is most typical of lymphangitic metastasis (46%), UIP, IPF (68%), collagen-vascular diseases such as rheumatoid lung disease and scleroderma (80%), and asbestosis (3,5,8,58,84,85,122,138,141). A #152201 Cust: LWW Au: Webb Pg. No. 101 101 TABLE 3-11 Predominance of Lung Disease on HRCT: Upper Lung Versus Lower Lung Lung disease Findings Upper Lung Sarcoidosis Nodules, fibrosis, conglomerate masses Silicosis Nodules, conglomerate masses Talcosis Conglomerate masses of fibrosis Chronic HP Mid- to upper-lung predominance typical Langerhans cell histiocytosis Reticulation in a few percent Pleuroparenchymal fibroelastosis Rare, reticulation traction bronchiectasis, honeycombing; pleural thickening Lower Lung Pulmonary edema Septal thickening Lymphangitic carcinoma and lymphoproliferative diseases Septal thickening UIP, IPF, collagen diseases, asbestosis Subpleural fibrosis; honeycombing NSIP Peripheral reticulation Other IPs Findings of fibrosis in a few basal predominance of abnormalities is typical of fibrotic NSIP (95). Pulmonary fibrosis of any cause has a basal predominance in approximately 60% of cases (84,85). Although HP may show an upper-lobe predominance, it more often appears to be diffuse or preponderant in the mid- (58,74) or lower lung zones (30%) (138). In a study by Silva et al. (137) comparing the HRCT findings in patients with IPF, NSIP, and chronic HP with fibrosis, a lower-zone predominance of abnormalities was more common in patients with IPF (83%) and NSIP (94%) than in those with chronic HP (31%) (p < 0.001). Also, although no significant difference was observed in the frequency of honeycombing in patients with chronic HP (64%) and IPF (67%), patients with IPF were more likely to have basal predominance of honeycombing (52%) than were those with chronic HP (11%) (p < 0.001). Upper-lobe fibrosis was seen in all patients with chronic HP, compared to 96% in IPF and 62% in NSIP (p < 0.001), and although an upper-zone predominance of abnormalities was uncommon, it was seen more frequently in patients with chronic HP (11%) than in those with IPF (2%) or NSIP (0%) (p = 0.02) (137). Uniform involvement of upper- and lower-lung zones was more common in patients with chronic HP (58%) than in those with IPF (15%) or NSIP (6%) (p < 0.001). Anterior Lung Versus Posterior Lung Some diseases produce their initial or most extensive abnormalities in the posterior lung (Table 3-12). The distinction between anterior and posterior, of course, is easily made on HRCT. However, it is important to DESIGN SERVICES OF 102 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease TABLE 3-12 Predominance of Lung Disease on HRCT: Posterior Lung Versus Anterior Lung Lung disease Findings Posterior lung UIP, NSIP Fibrosis Asbestosis Fibrosis Collagen-vascular disease Fibrosis Silicosis Fibrosis; conglomerate masses Sarcoidosis Fibrosis; conglomerate masses Pulmonary edema Septal thickening Lymphangitic carcinoma and lymphoproliferative disease Septal thickening HP Findings of fibrosis Anterior lung Post-ARDS fibrosis Subpleural fibrosis; honeycombing; traction bronchiectasis Radiation fibrosis in some patients (e.g., those with breast cancer) Reticulation; volume loss; traction bronchiectasis; honeycombing in some recognize the value of using both prone and supine scans in this regard. Areas of increased attenuation that are limited to the posterior lung on scans obtained in the supine position can reflect normal dependent volume loss; prone scans are essential in making a confident diagnosis of early posterior lung disease. Although the percentages vary in different series, a posterior preponderance of disease is particularly common in scleroderma (60%), sarcoidosis (32%–36%), silicosis (31%–38%) HP (23%), IPF (9%–21%) and other causes of UIP, and NSIP (50,84,85,138,145). A posterior predominance of abnormalities is also common in patients with asbestosis, lymphangitic carcinomatosis, and pulmonary edema (3,8,84,85,122,138,141,145). In patients with pulmonary edema, the predominant abnormality is more appropriately referred to as dependent rather than posterior. An anterior predominance of lung disease is unusual but has been reported in adult survivors of ARDS (78). In this study, HRCT was obtained during the acute illness and at follow-up in 27 patients with ARDS. At follow-up CT, a reticular pattern was the most prevalent abnormality (85%), with a striking anterior distribution. This finding was related to the duration of mechanical ventilation and was inversely correlated with the extent of parenchymal opacification on scans obtained during the acute illness. Anterior lung fibrosis is common in patients having radiation therapy for breast cancer. Unilateral Disease Versus Bilateral Disease A unilateral predominance of abnormalities is most typical of lymphangitic spread of carcinoma, which is often asymmetric in distribution; this was seen in nearly 40% #152201 Cust: LWW Au: Webb Pg. No. 102 of patients with lymphangitic spread of carcinoma in one series (84). Asymmetry or unilateral predominance of findings is also common in patients with sarcoidosis, ranging from 9% to 21%. It is somewhat less frequent in association with silicosis (2%–21%), IPF (3%–14%), and HP (5%) (84,85). Drug reactions resulting in lung fibrosis or other abnormalities may appear asymmetrical or predominantly unilateral. REFERENCES 1. Zerhouni E. Computed tomography of the pulmonary p arenchyma: an overview. Chest 1989;95:901–907. 2.Webb WR. High-resolution CT of the lung parenchyma. Radiol Clin North Am 1989;27:1085–1097. 3. Swensen SJ, Aughenbaugh GL, Brown LR. High-resolution computed tomography of the lung. Mayo Clin Proc 1989;64:1284–1294. 4. Stein MG, Mayo J, Müller N, et al. Pulmonary lymphangitic spread of carcinoma: appearance on CT scans. Radiology 1987;162: 371–375. 5. Müller NL, Miller RR, Webb WR, et al. Fibrosing alveolitis: CTpathologic correlation. Radiology 1986;160:585–588. 6. Murata K, Itoh H, Todo G, et al. Centrilobular lesions of the lung: demonstration by high-resolution CT and pathologic correlation. Radiology 1986;161:641–645. 7.Munk PL, Müller NL, Miller RR, et al. Pulmonary lymphangitic carcinomatosis: CT and pathologic findings. Radiology 1988;166:705–709. 8. Aberle DR, Gamsu G, Ray CS, et al. Asbestos-related pleural and parenchymal fibrosis: detection with high-resolution CT. Radiology 1988;166:729–734. 9. Austin JH, Müller NL, Friedman PJ, et al. Glossary of terms for CT of the lungs: recommendations of the nomenclature committee of the Fleischner society. Radiology 1996;200:327–331. 10.Hansell DM, Bankier AA, MacMahon H, et al. Fleischner society: glossary of terms for thoracic imaging. Radiology 2008;246: 697–722. 11. Akira M, Yamamoto S, Yokoyama K, et al. Asbestosis: high-resolution CT-pathologic correlation. Radiology 1990;176:389–394. 12. Zerhouni EA, Naidich DP, Stitik FP, et al. Computed tomography of the pulmonary parenchyma: part 2. Interstitial disease. J Thorac Imaging 1985;1:54–64. 13. Bergin CJ, Coblentz CL, Chiles C, et al. Chronic lung diseases: specific diagnosis using CT. AJR Am J Roentgenol 1989;152:1183–1188. 14. Webb WR, Müller NL, Naidich DP. Standardized terms for highresolution computed tomography of the lung: a proposed glossary. J Thorac Imaging 1993;8:167–185. 15. Sumikawa H, Johkoh T, Ichikado K, et al. Usual interstitial pneumonia and chronic idiopathic interstitial pneumonia: analysis of CT appearance in 92 patients. Radiology 2006;241:258–266. 16.Sibtain NA, Ujita M, Wilson R, et al. Interlobular septal thickening in idiopathic bronchiectasis: a thin-section CT study of 94 patients. Radiology 2005;237:1091–1096. 17. Copley SJ, Wells AU, Hawtin KE, et al. Lung morphology in the elderly: comparative CT study of subjects over 75 years old versus those under 55 years old. Radiology 2009;251:566–573. 18. Kang EY, Grenier P, Laurent F, et al. Interlobular septal thickening: patterns at high-resolution computed tomography. J Thorac Imaging 1996;11:260–264. 19. Webb WR. Thin-section CT of the secondary pulmonary lobule: anatomy and the image—the 2004 Fleischner lecture. Radiology 2006;239:322–338. 20.Andreu J, Hidalgo A, Pallisa E, et al. Septal thickening: HRCT findings and differential diagnosis. Curr Probl Diagn Radiol 2004;33:226–237. 21. Collins J. CT signs and patterns of lung disease. Radiol Clin North Am 2001;39:1115–1135. DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities 22. Cassart M, Genevois PA, Kramer M, et al. Pulmonary venoocclusive disease: CT findings before and after single-lung transplantation. AJR Am J Roentgenol 1993;160:759–760. 23.Storto ML, Kee ST, Golden JA, et al. Hydrostatic pulmonary edema: high-resolution CT findings. AJR Am J Roentgenol 1995;165:817–820. 24. Swensen SJ, Tashjian JH, Myers JL, et al. Pulmonary venoocclusive disease: CT findings in eight patients. AJR Am J Roentgenol 1996;167:937–940. 25.Franquet T, Müller NL, Lee KS, et al. High-resolution CT and pathologic findings of noninfectious pulmonary complications after hematopoietic stem cell transplantation. AJR Am J Roentgenol 2005;184:629–637. 26. Dufour B, Maitre S, Humbert M, et al. High-resolution CT of the chest in four patients with pulmonary capillary hemangiomatosis or pulmonary venoocclusive disease. AJR Am J Roentgenol 1998;171:1321–1324. 27. Resten A, Maitre S, Humbert M, et al. Pulmonary hypertension: CT of the chest in pulmonary venoocclusive disease. AJR Am J Roentgenol 2004;183:65–70. 28. Ren H, Hruban RH, Kuhlman JE, et al. Computed tomography of inflation-fixed lungs: the beaded septum sign of pulmonary metastases. J Comput Assist Tomogr 1989;13:411–416. 29. Lynch DA, Hay T, Newell JD Jr, et al. Pediatric diffuse lung disease: diagnosis and classification using high-resolution CT. AJR Am J Roentgenol 1999;173:713–718. 30. Colby TV, Swensen SJ. Anatomic distribution and histopathologic patterns in diffuse lung disease: correlation with HRCT. J Thorac Imaging 1996;11:1–26. 31. Nobre LF, Müller NL, de Souza Junior AS, et al. Congenital pulmonary lymphangiectasia: CT and pathologic findings. J Thorac Imaging 2004;19:56–59. 32. Raman SP, Pipavath SNJ, Raghu G, et al. Imaging of thoracic lymphatic diseases. AJR Am J Roentgenol 2009;193:1504–1513. 33.Graham CM, Stern EJ, Finkbeiner WE, et al. High-resolution CT appearance of diffuse alveolar septal amyloidosis. AJR Am J Roentgenol 1992;158:265–267. 34.Wittenberg KH, Swensen SJ, Myers JL. Pulmonary involvement with Erdheim-Chester disease: radiographic and CT findings. AJR Am J Roentgenol 2000;174:1327–1331. 35.Franquet T. Imaging of pulmonary viral pneumonia. Radiology 2011;260:18–39. 36. Ujita M, Renzoni EA, Veeraraghavan S, et al. Organizing pneumonia: perilobular pattern at thin-section CT. Radiology 2004;232:757–761. 37.Murch CR, Carr DH. Computed tomography appearances of pulmonary alveolar proteinosis. Clin Radiol 1989;40:240–243. 38.Godwin JD, Müller NL, Takasugi JE. Pulmonary alveolar proteinosis: CT findings. Radiology 1988;169:609–613. 39. Lee KN, Levin DL, Webb WR, et al. Pulmonary alveolar proteinosis: high-resolution CT, chest radiographic, and functional correlations. Chest 1997;111:989–995. 40.Murayama S, Murakami J, Yabuuchi H, et al. “Crazy paving appearance” on high resolution CT in various diseases. J Comput Assist Tomogr 1999;23:749–752. 41.Johkoh T, Itoh H, Müller NL, et al. Crazy-paving appearance at thin-section CT: spectrum of disease and pathologic findings. Radiology 1999;211:155–160. 42. Rossi SE, Erasmus JJ, Volpacchio M, et al. “Crazy-paving” pattern at thin-section CT of the lungs: radiologic-pathologic overview. Radiographics 2003;23:1509–1519. 43.Johkoh T, Müller NL, Pickford HA, et al. Lymphocytic interstitial pneumonia: thin-section CT findings in 22 patients. Radiology 1999;212:567–572. 44. Ichikawa Y, Kinoshita M, Koga T, et al. Lung cyst formation in Lymphocytic interstitial pneumonia: CT features. J Comput Assist Tomogr 1994;18:745–748. 45. McGuinness G, Scholes JV, Jagirdar JS, et al. Unusual lymphoproliferative disorders in nine adults with HIV or AIDS: CT and pathologic findings. Radiology 1995;197:59–65. #152201 Cust: LWW Au: Webb Pg. No. 103 103 46. Lynch DA, Webb WR, Gamsu G, et al. Computed tomography in pulmonary sarcoidosis. J Comput Assist Tomogr 1989;13:405–410. 47.Remy-Jardin M, Beuscart R, Sault MC, et al. Subpleural micronodules in diffuse infiltrative lung diseases: evaluation with thin-section CT scans. Radiology 1990;177:133–139. 48.Müller NL, Kullnig P, Miller RR. The CT findings of pulmonary sarcoidosis: analysis of 25 patients. AJR Am J Roentgenol 1989;152:1179–1182. 49.Traill ZC, Maskell GF, Gleeson FV. High-resolution CT findings of pulmonary sarcoidosis. AJR Am J Roentgenol 1997;168: 1557–1560. 50.Remy-Jardin M, Degreef JM, Beuscart R, et al. Coal worker’s pneumoconiosis: CT assessment in exposed workers and correlation with radiographic findings. Radiology 1990;177:363–371. 51. Pickford HA, Swensen SJ, Utz JP. Thoracic cross-sectional imaging of amyloidosis. AJR Am J Roentgenol 1997;168:351–355. 52. Webb WR, Stein MG, Finkbeiner WE, et al. Normal and diseased isolated lungs: high-resolution CT. Radiology 1988;166:81–87. 53.Souza AS Jr, Müller NL, Marchiori E, et al. Pulmonary abnormalities in ankylosing spondylitis: inspiratory and expiratory high-resolution CT findings in 17 patients. J Thorac Imaging 2004;19:259–263. 54. Remy-Jardin M, Sobaszek A, Duhamel A, et al. Asbestos-related pleuropulmonary diseases: evaluation with low-dose four-detector row spiral CT. Radiology 2004;233:182–190. 55.Turetschek K, Ebner W, Fleischmann D, et al. Early pulmonary involvement in ankylosing spondylitis: assessment with thin- section CT. Clin Radiol 2000;55:632–636. 56. Afeltra A, Zennaro D, Garzia P, et al. Prevalence of interstitial lung involvement in patients with connective tissue diseases assessed with high-resolution computed tomography. Scand J Rheumatol 2006;35:388–394. 57. Nishimura K, Kitaichi M, Izumi T, et al. Usual interstitial pneumonia: histologic correlation with high-resolution CT. Radiology 1992;182:337–342. 58.Primack SL, Hartman TE, Hansell DM, et al. End-stage lung disease: CT findings in 61 patients. Radiology 1993;189:681–686. 59. Murata K, Khan A, Herman PG. Pulmonary parenchymal disease: evaluation with high-resolution CT. Radiology 1989;170:629–635. 60.Johkoh T, Müller NL, Ichikado K, et al. Perilobular pulmonary opacities: high-resolution CT findings and pathologic correlation. J Thorac Imaging 1999;14:172–177. 61.Arakawa H, Honma K. Honeycomb lung: history and current concepts. AJR Am J Roentgenol 2011;196:773–782. 62.Genereux GP. The end-stage lung: pathogenesis, pathology, and radiology. Radiology 1975;116:279–289. 63.Raghu G, Collard HR, Egan JJ, et al. An official ATS/ERS/JRS/ ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 2011;183:788–824. 64.Lynch DA, Travis WD, Müller NL, et al. Idiopathic interstitial pneumonias: CT features. Radiology 2005;236:10–21. 65.Johkoh T, Müller NL, Colby TV, et al. Nonspecific interstitial pneumonia: correlation between thin-section CT findings and pathologic subgroups in 55 patients. Radiology 2002;225:199–204. 66. Kim EA, Lee KS, Johkoh T, et al. Interstitial lung diseases associated with collagen vascular diseases: radiologic and histopathologic findings. Radiographics 2002;22 Spec No:S151–S165. 67. Tanaka N, Kim JS, Newell JD, et al. Rheumatoid arthritis-related lung diseases: CT findings. Radiology 2004;232:81–91. 68.Seely JM, Jones LT, Wallace C, et al. Systemic sclerosis: using high-resolution CT to detect lung disease in children. AJR Am J Roentgenol 1998;170:691–697. 69. Akira M, Yamamoto S, Inoue Y, et al. High-resolution CT of asbestosis and idiopathic pulmonary fibrosis. AJR Am J Roentgenol 2003;181:163–169. 70. Copley SJ, Wells AU, Sivakumaran P, et al. Asbestosis and i diopathic pulmonary fibrosis: comparison of thin-section CT features. Radiology 2003;229:731–736. DESIGN SERVICES OF 104 s e c t i o n II Approach to HRCT Diagnosis and Findings of Lung Disease 71. Attili AK, Kazerooni EA, Gross BH, et al. Smoking-related interstitial lung disease: radiologic-clinical-pathologic correlation. Radiographics 2008;28:1383–1396. 72.Galvin JR, Franks TJ. Smoking-related lung disease. J Thorac Imaging 2009;24:274–284. 73. Johkoh T, Müller NL, Cartier Y, et al. Idiopathic interstitial pneumonias: diagnostic accuracy of thin-section CT in 129 patients. Radiology 1999;211:555–560. 74.Adler BD, Padley SP, Müller NL, et al. Chronic hypersensitivity pneumonitis: high-resolution CT and radiographic features in 16 patients. Radiology 1992;185:91–95. 75. Kim TS, Lee KS, Chung MP, et al. Nonspecific interstitial pneumonia with fibrosis: high-resolution CT and pathologic findings. AJR Am J Roentgenol 1998;171:1645–1650. 76.Park JS, Lee KS, Kim JS, et al. Nonspecific interstitial pneumonia with fibrosis: radiographic and CT findings in seven patients. Radiology 1995;195:645–648. 77.Kim EY, Lee KS, Chung MP, et al. Nonspecific interstitial pneumonia with fibrosis: serial high-resolution CT findings with functional correlation. AJR Am J Roentgenol 1999;173:949–953. 78. Desai SR, Wells AU, Rubens MB, et al. Acute respiratory distress syndrome: CT abnormalities at long-term follow-up. Radiology 1999;210:29–35. 79. Remy-Jardin M, Giraud F, Remy J, et al. Importance of groundglass attenuation in chronic diffuse infiltrative lung disease: pathologic-CT correlation. Radiology 1993;189:693–698. 80.Wells AU, Hansell DM, Rubens MB, et al. The predictive value of appearances of thin-section computed tomography in fibrosing alveolitis. Am Rev Respir Dis 1993;148:1076–1082. 81. American Thoracic Society/European Respiratory Society international multidisciplinary consensus classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 2002;165:277–304. 82.Tung KT, Wells AU, Rubens MB, et al. Accuracy of the typical computed tomographic appearances of fibrosing alveolitis. Thorax 1993;48:334–338. 83. Al-Jarad N, Strickland B, Pearson MC, et al. High-resolution computed tomographic assessment of asbestosis and cryptogenic fibrosing alveolitis: a comparative study. Thorax 1992;47:645–650. 84. Mathieson JR, Mayo JR, Staples CA, et al. Chronic diffuse infiltrative lung disease: comparison of diagnostic accuracy of CT and chest radiography. Radiology 1989;171:111–116. 85. Grenier P, Valeyre D, Cluzel P, et al. Chronic diffuse interstitial lung disease: diagnostic value of chest radiography and high-resolution CT. Radiology 1991;179:123–132. 86. Padley SPG, Hansell DM, Flower CDR, et al. Comparative accuracy of high resolution computed tomography and chest radiography in the diagnosis of chronic diffuse infiltrative lung disease. Clin Radiol 1991;44:222–226. 87. Nishimura K, Izumi T, Kitaichi M, et al. The diagnostic accuracy of high-resolution computed tomography in diffuse infiltrative lung diseases. Chest 1993;104:1149–1155. 88.Swensen SJ, Aughenbaugh GL, Myers JL. Diffuse lung disease: diagnostic accuracy of CT in patients undergoing surgical biopsy of the lung. Radiology 1997;205:229–234. 89. Hunninghake GW, Lynch DA, Galvin JR, et al. Radiologic findings are strongly associated with a pathologic diagnosis of usual interstitial pneumonia. Chest 2003;124:1215–1223. 90. Hunninghake GW, Zimmerman MB, Schwartz DA, et al. Utility of a lung biopsy for the diagnosis of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 2001;164:193–196. 91. Iwasawa T, Asakura A, Sakai F, et al. Assessment of prognosis of patients with idiopathic pulmonary fibrosis by computer-aided analysis of CT images. J Thorac Imaging 2009;24:216–222. 92.Hwang J-H, Misumi S, Curran-Everett D, et al. Longitudinal follow-up of fibrosing interstitial pneumonia: relationship between physiologic testing, computed tomography changes, and survival rate. J Thorac Imaging 2011;26:209–217. #152201 Cust: LWW Au: Webb Pg. No. 104 93.Watadani T, Sakai F, Johkoh T, et al. Interobserver variability in the CT assessment of honeycombing in the lungs. Radiology 2013;266:936–944. 94. Kligerman SJ, Groshong S, Brown KK, et al. Nonspecific interstitial pneumonia: radiologic, clinical, and pathologic considerations. Radiographics 2009;29:73–87. 95. Travis WD, Hunninghake G, King TE Jr, et al. Idiopathic nonspecific interstitial pneumonia: report of an american thoracic society project. Am J Respir Crit Care Med 2008;177:1338–1347. 96.Akira M. High-resolution CT in the evaluation of occupational and environmental disease. Radiol Clin North Am 2002;40:43–59. 97. Akira M, Ishikawa H, Yamamoto S. Drug-induced pneumonitis: thinsection CT findings in 60 patients. Radiology 2002;224:852–860. 98. Westcott JL, Cole SR. Traction bronchiectasis in end-stage pulmonary fibrosis. Radiology 1986;161:665–669. 99. Logan PM. Thoracic manifestations of external beam radiotherapy. AJR Am J Roentgenol 1998;171:569–577. 100.Murata K, Takahashi M, Mori M, et al. Peribronchovascular interstitium of the pulmonary hilum: normal and abnormal findings on thin-section electron-beam CT. AJR Am J Roentgenol 1996;166:309–312. 101.Weibel ER. Looking into the lung: what can it tell us? AJR Am J Roentgenol 1979;133:1021–1031. 102.Bergin CJ, Müller NL. CT in the diagnosis of interstitial lung disease. AJR Am J Roentgenol 1985;145:505–510. 103. Johkoh T, Ikezoe J, Tomiyama N, et al. CT findings in lymphangitic carcinomatosis of the lung: correlation with histologic findings and pulmonary function tests. AJR Am J Roentgenol 1992;158:1217–1222. 104.Lee KS, Kim Y, Primack SL. Imaging of pulmonary lymphomas. AJR Am J Roentgenol 1997;168:339–345. 105.Heyneman LE, Johkoh T, Ward S, et al. Pulmonary leukemic infiltrates: high-resolution CT findings in 10 patients. AJR Am J Roentgenol 2000;174:517–521. 106. Bessis L, Callard P, Gotheil C, et al. High-resolution CT of parenchymal lung disease: precise correlation with histologic findings. Radiographics 1992;12:45–58. 107.Criado E, Sanchez M, Ramirez J, et al. Pulmonary sarcoidosis: typical and atypical manifestations at high-resolution CT with pathologic correlation. Radiographics 2010;30:1567–1586. 108.Wintermark M, Wicky S, Schnyder P, et al. Blunt traumatic pneumomediastinum: using CT to reveal the Macklin effect. AJR Am J Roentgenol 1999;172:129–130. 109. Kemper AC, Steinberg KP, Stern EJ. Pulmonary interstitial emphysema: CT findings. AJR Am J Roentgenol 1999;172:1642. 110. Satoh K, Kobayashi T, Kawase Y, et al. CT appearance of interstitial pulmonary emphysema. J Thorac Imaging 1996;11:153–154. 111. Chong S, Lee KS, Chung MJ, et al. Pneumoconiosis: comparison of imaging and pathologic findings. Radiographics 2006;26:59–77. 112.Weibel ER, Taylor CR. Design and structure of the human lung. In: Fishman AP, ed. Pulmonary diseases and disorders. 2nd ed. New York, NY: McGraw-Hill; 1988:11–60. 113.Matsuoka S, Uchiyama K, Shima H, et al. Bronchoarterial ratio and bronchial wall thickness on high-resolution CT in asymp tomatic subjects: correlation with age and smoking. AJR Am J Roentgenol 2003;180:513–518. 114. Awadh N, Müller NL, Park CS, et al. Airway wall thickness in patients with near fatal asthma and control groups: assessment with high resolution computed tomographic scanning. Thorax 1998;53:248–253. 115. Bankier AA, Fleischmann D, De Maertelaer V, et al. Subjective differentiation of normal and pathological bronchi on thin-section CT: impact of observer training. Eur Respir J 1999;13:781–786. 116. Naidich DP, McCauley DI, Khouri NF, et al. Computed tomography of bronchiectasis. J Comput Assist Tomogr 1982;6:437–444. 117. Grenier P, Maurice F, Musset D, et al. Bronchiectasis: assessment by thin-section CT. Radiology 1986;161:95–99. 118.Munro NC, Cooke JC, Currie DC, et al. Comparison of thin section computed tomography with bronchography for identifying DESIGN SERVICES OF C hapter 3 HRCT Findings: Linear and Reticular Opacities bronchiectatic segments in patients with chronis sputum production. Thorax 1990;45:135–139. 119. Grenier P, Lenoir S, Brauner M. Computed tomographic assessment of bronchiectasis. Semin Ultrasound CT MR 1990;11:430–441. 120. Neeld DA, Goodman LR, Gurney JW, et al. Computerized tomography in the evaluation of allergic bronchopulmonary aspergillosis. Am Rev Respir Dis 1990;142:1200–1206. 121.Todo G, Herman PG. High-resolution computed tomography of the pig lung. Invest Radiol 1986;21:689–696. 122.Aberle DR, Gamsu G, Ray CS. High-resolution CT of benign asbestos-related diseases: clinical and radiographic correlation. AJR Am J Roentgenol 1988;151:883–891. 123.Lynch DA, Gamsu G, Ray CS, et al. Asbestos-related focal lung masses: manifestations on conventional and high-resolution CT scans. Radiology 1988;169:603–607. 124.Im JG, Itoh H, Shim YS, et al. Pulmonary tuberculosis: CT findings—early active disease and sequential change with antituberculous therapy. Radiology 1993;186:653–660. 125. Altin R, Ozdolap S, Savranlar A, et al. Comparison of early and late pleuropulmonary findings of ankylosing spondylitis by highresolution computed tomography and effects on patients’ daily life. Clin Rheumatol 2005;24:22–28. 126.Kiris A, Ozgocmen S, Kocakoc E, et al. Lung findings on high resolution CT in early ankylosing spondylitis. Eur J Radiol 2003; 47:71–76. 127.Senocak O, Manisali M, Ozaksoy D, et al. Lung parenchyma changes in ankylosing spondylitis: demonstration with high resolution CT and correlation with disease duration. Eur J Radiol 2003;45:117–122. 128. Yoshimura H, Hatakeyama M, Otsuji H, et al. Pulmonary asbestosis: CT study of subpleural curvilinear shadow. Work in progress. Radiology 1986;158:653–658. 129. Schurawitzki H, Stiglbauer R, Graninger W, et al. Interstitial lung disease in progressive systemic sclerosis: high-resolution CT versus radiography. Radiology 1990;176:755–759. 130. Swensen SJ, Aughenbaugh GL, Douglas WW, et al. High-resolution CT of the lungs: findings in various pulmonary diseases. AJR Am J Roentgenol 1992;158:971–979. 131.Otake S, Takahashi M, Ishigaki T. Focal pulmonary interstitial opacities adjacent to thoracic spine osteophytes. AJR Am J Roentgenol 2002;179:893–896. #152201 Cust: LWW Au: Webb Pg. No. 105 105 132.Morimoto S, Takeuchi N, Imanaka H, et al. Gravity dependent atelectasis: radiologic, physiologic and pathologic correlation in rabbits on high-frequency ventilation. Invest Radiol 1989;24:522–530. 133.Lee KN, Yoon SK, Sohn CH, et al. Dependent lung opacity at thin-section CT: evaluation by spirometrically-gated CT of the influence of lung volume. Korean J Radiol 2002;3:24–29. 134.Kubota H, Hosoya T, Kato M, et al. Plate-like atelectasis at the corticomedullary junction of the lung: CT observation and hypothesis. Radiat Med 1983;1:305–310. 135. Bergin CJ, Müller NL. CT of interstitial lung disease: a diagnostic approach. AJR Am J Roentgenol 1987;148:9–15. 136.Gurney JW. Cross-sectional physiology of the lung. Radiology 1991;178:1–10. 137.Silva CIS, Müller NL, Lynch DA, et al. Chronic hypersensitivity pneumonitis: differentiation from idiopathic pulmonary fibrosis and nonspecific interstitial pneumonia by using thin-section CT. Radiology 2008;246:288–297. 138. Grenier P, Chevret S, Beigelman C, et al. Chronic diffuse infiltrative lung disease: determination of the diagnostic value of clincial data, chest radiography, and CT with bayesian analysis. Radiology 1994;1994:383–390. 139. Müller NL, Staples CA, Miller RR. Bronchiolitis obliterans organizing pneumonia: CT features in 14 patients. AJR Am J Roentgenol 1990;154:983–987. 140. Hartman TE, Primack SL, Swensen SJ, et al. Desquamative interstitial pneumonia: thin-section CT findings in 22 patients. Radiology 1993;187:787–790. 141. Remy-Jardin M, Remy J, Wallaert B, et al. Pulmonary involvement in progressive systemic sclerosis: sequential evaluation with CT, pulmonary function tests, and bronchoalveolar lavage. Radiology 1993;188:499–506. 142.Gurney JW, Schroeder BA. Upper lobe disease: physiological correlates. Radiology 1988;167:359–366. istiocytosis 143. Brauner MW, Grenier P, Mouelhi MM, et al. Pulmonary h X: evaluation with high resolution CT. Radiology 1989;172:255–258. 144. Moore AD, Godwin JD, Müller NL, et al. Pulmonary histiocytosis X: comparison of radiographic and CT findings. Radiology 1989;172:249–254. 145.Brauner MW, Grenier P, Mompoint D, et al. Pulmonary sarcoidosis: evaluation with high-resolution CT. Radiology 1989; 172:467–471. DESIGN SERVICES OF